Brief HPI:
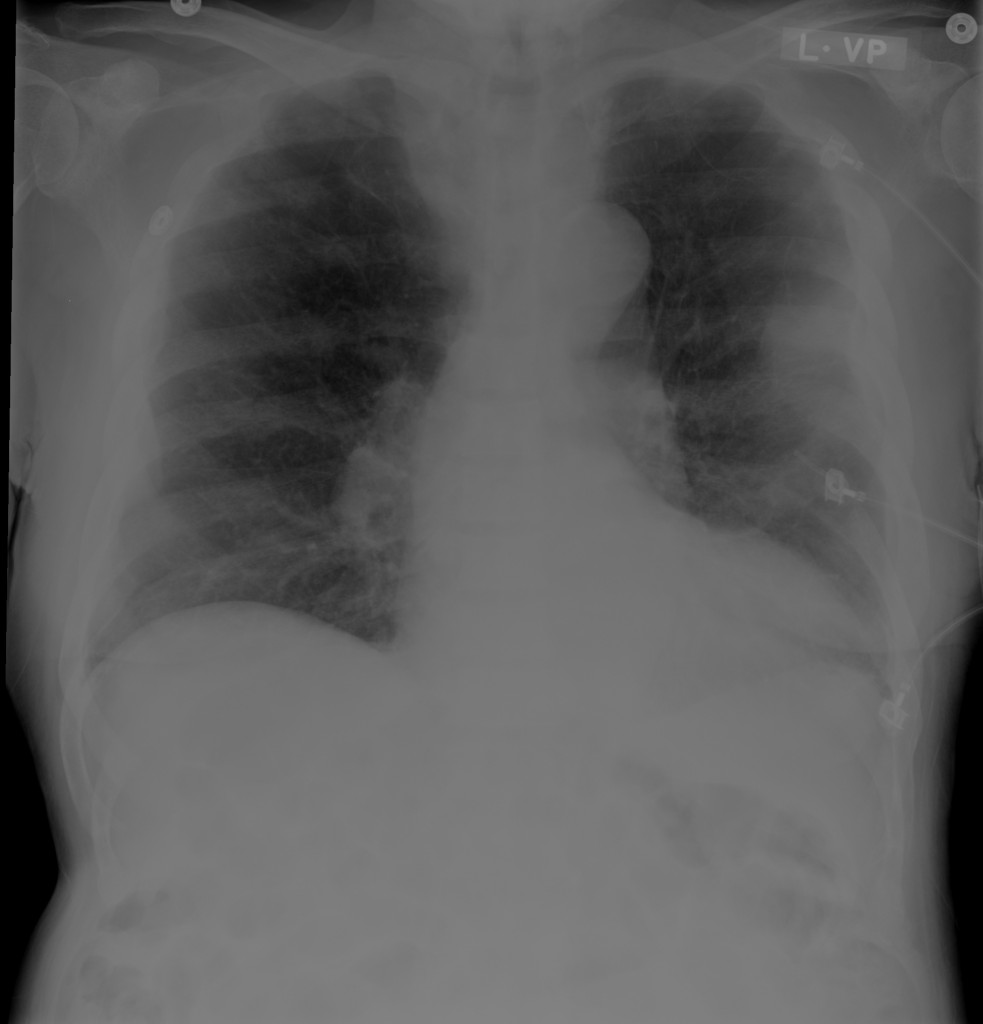
An approximately 80-year-old male with unknown medical history is brought to the emergency department from a skilled nursing facility after unwitnessed arrest – EMS providers established return of spontaneous circulation after chest compressions and epinephrine. On arrival, the patient was hypotensive (MAP 40mmHg) and hypoxic (SpO2 85%) with mask ventilation. The patient was intubated, resuscitated with intravenous fluids and started on vasopressors. Imaging demonstrated lung consolidation consistent with multifocal pneumonia versus aspiration. Laboratory studies were obtained:
- CBC: WBC: 49.2 (N: 64%, Bands: 20%)
- ABG: pH: 7.07, pCO2: 73mmHg
- Lactate: 9.1mmol/L
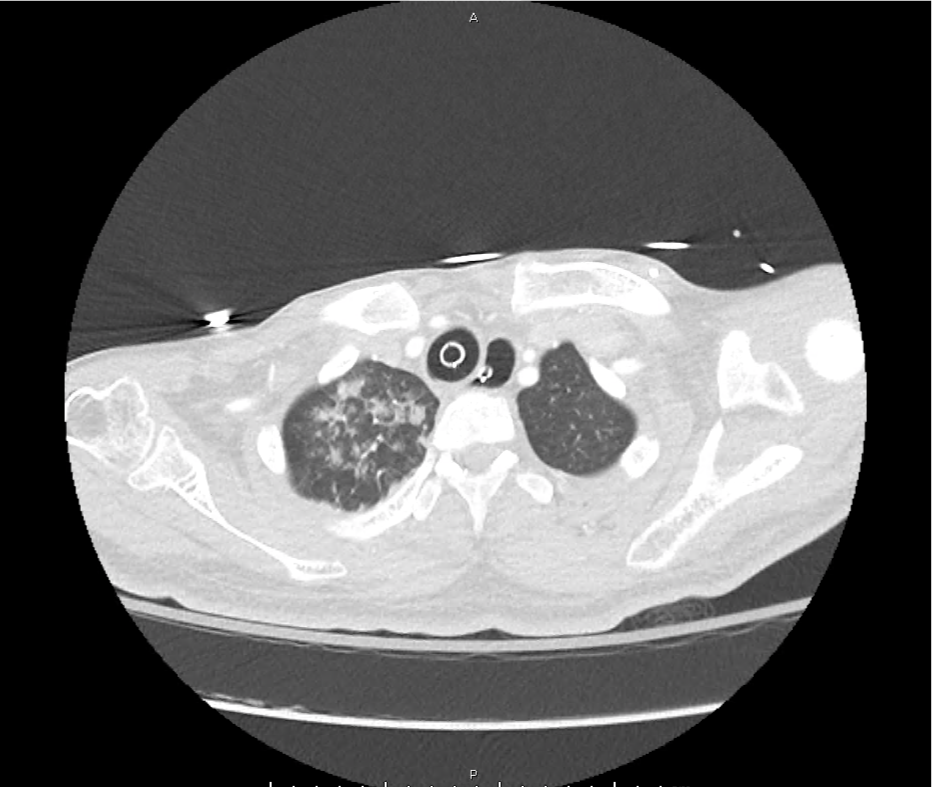
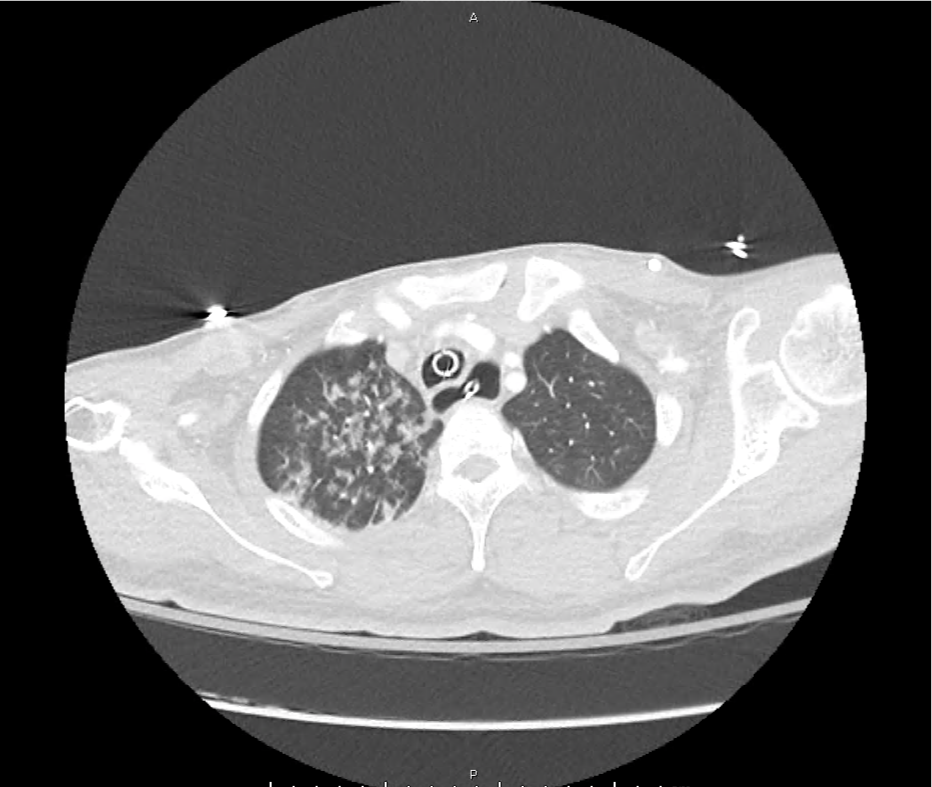
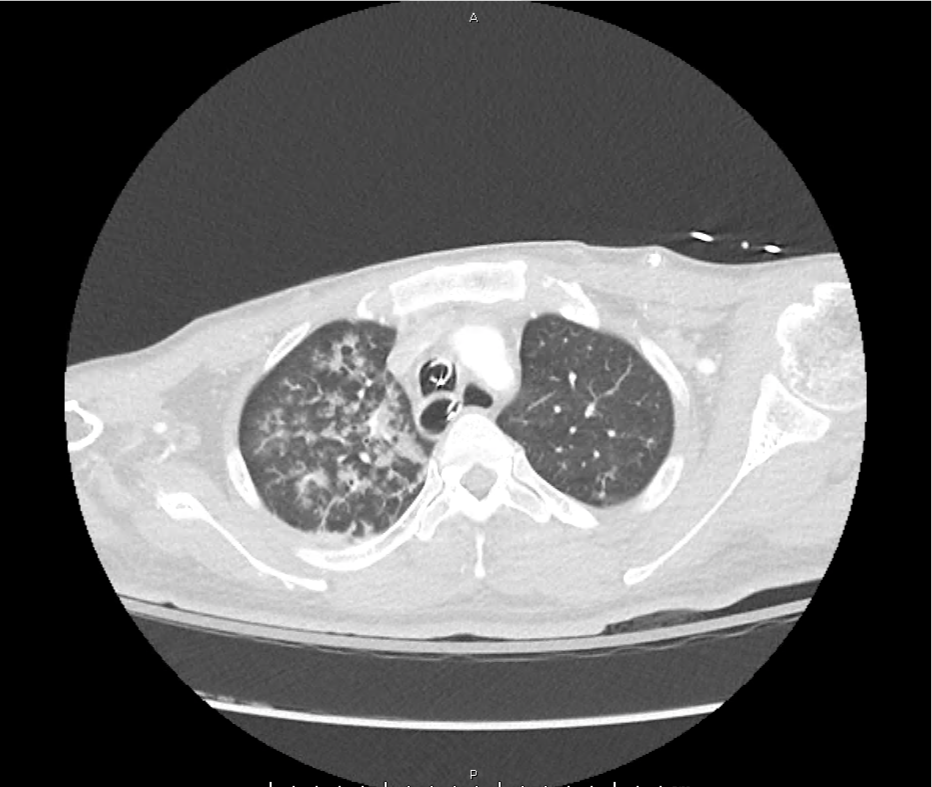
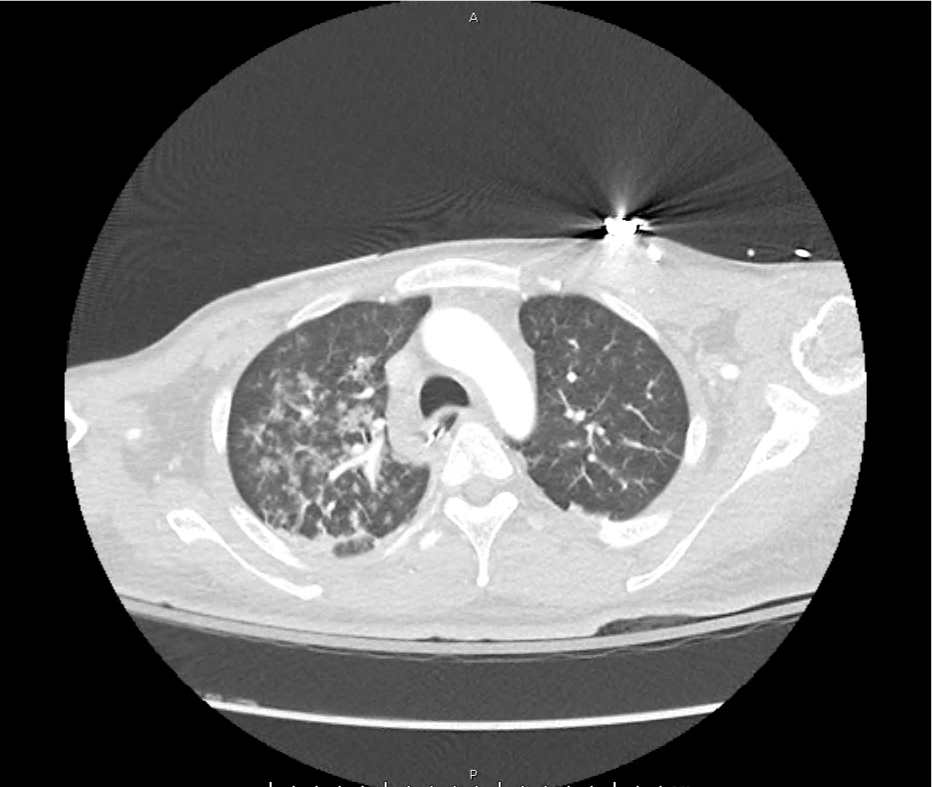
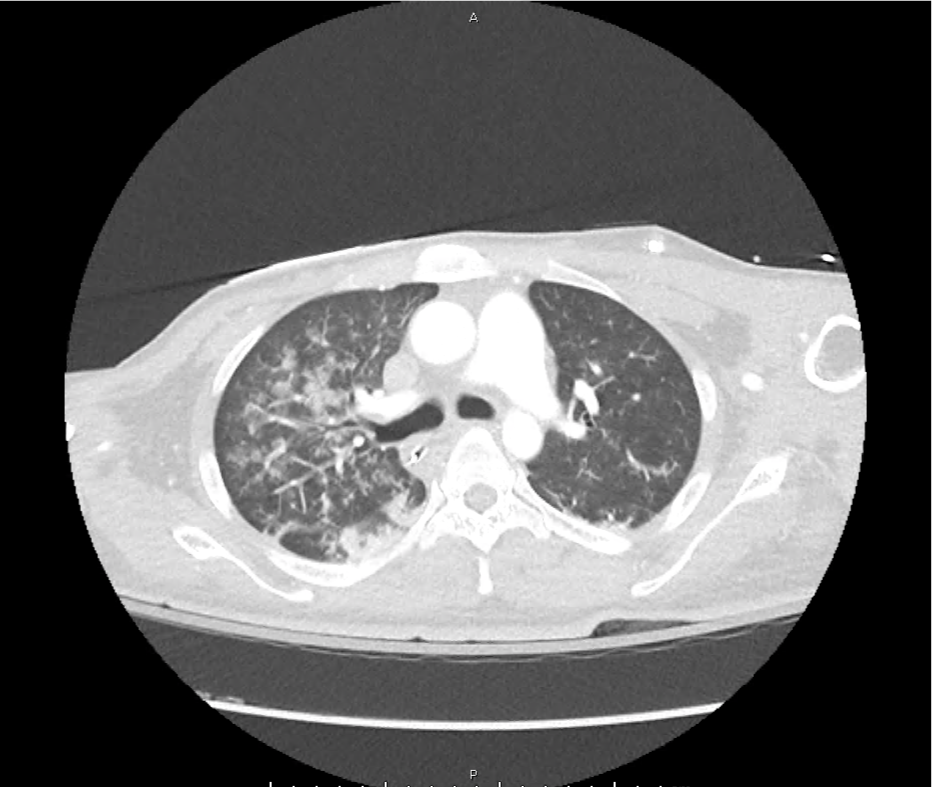
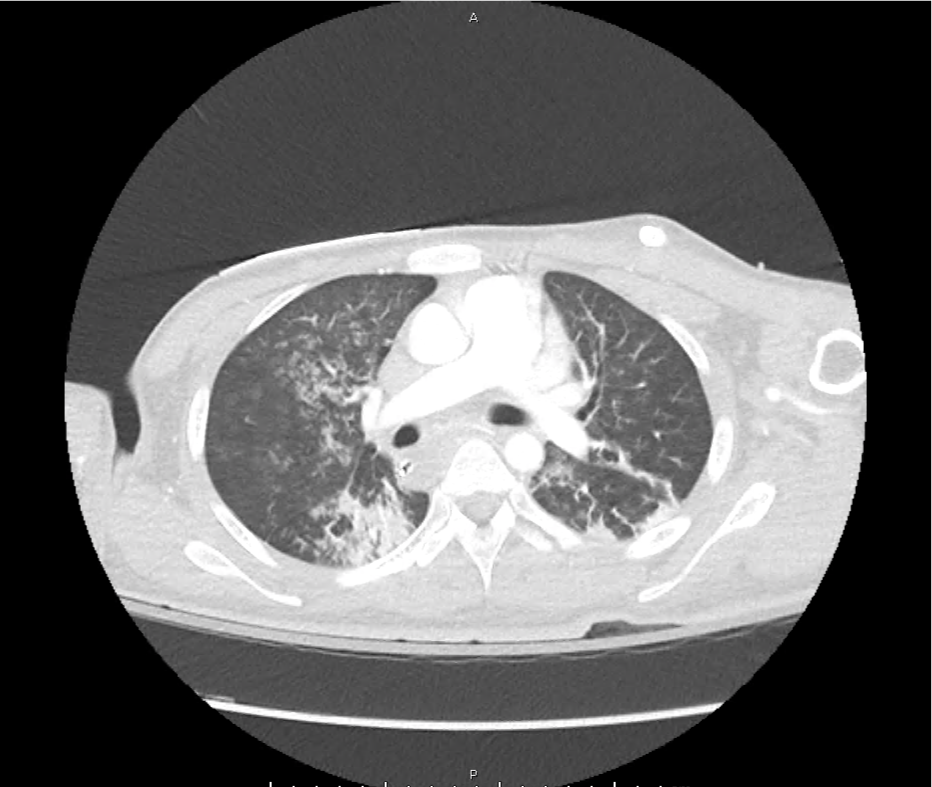
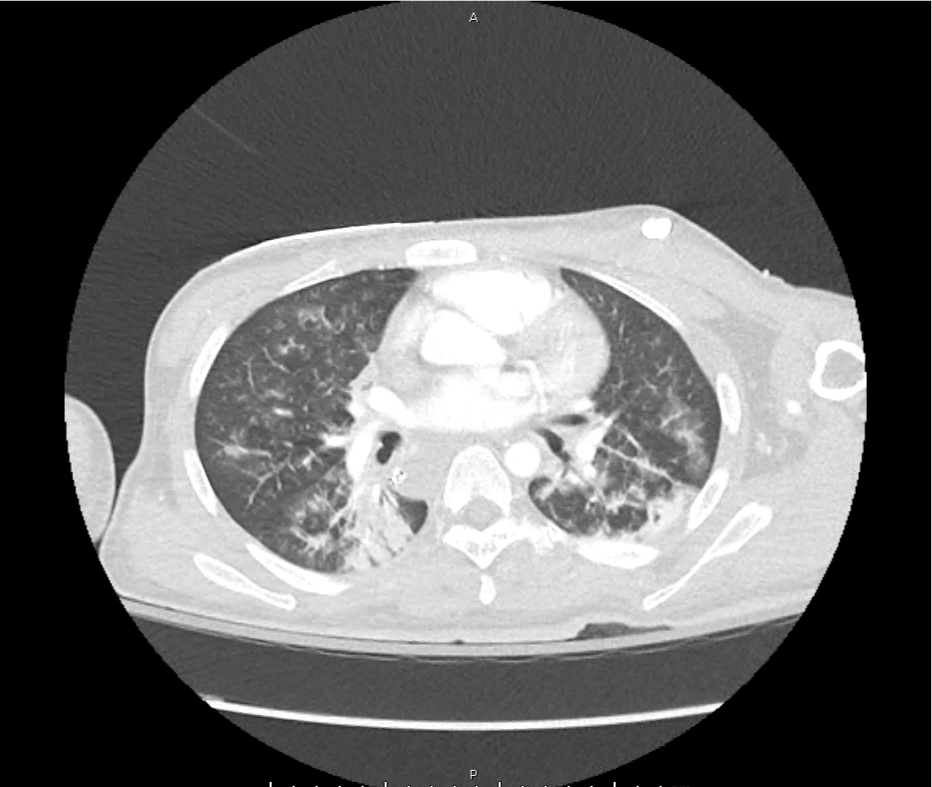
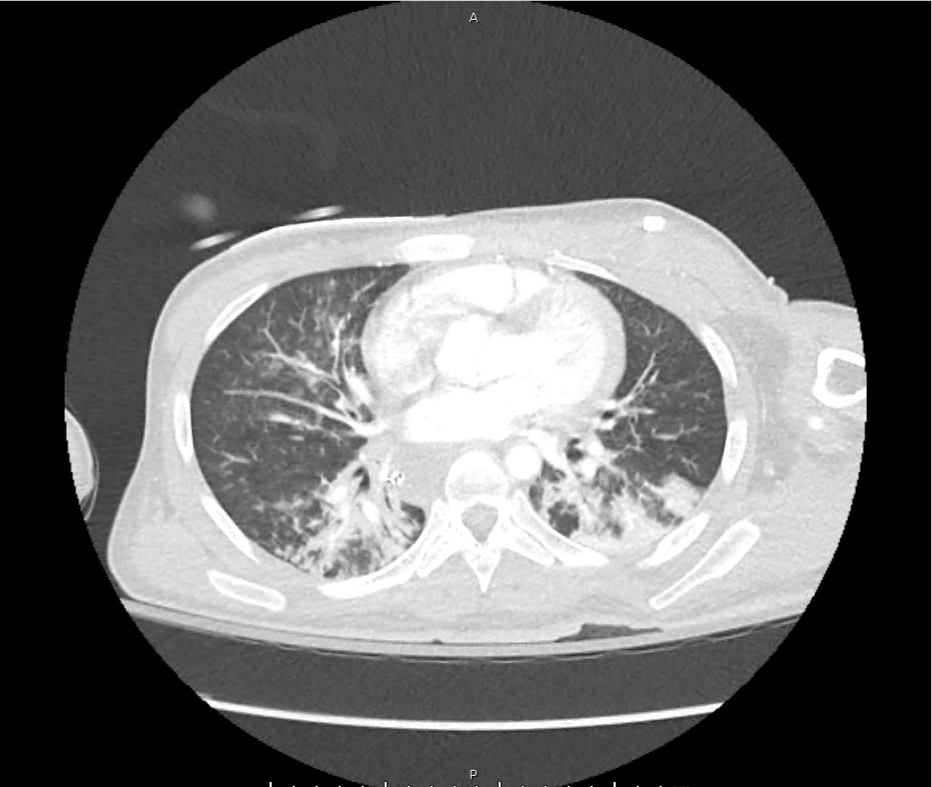
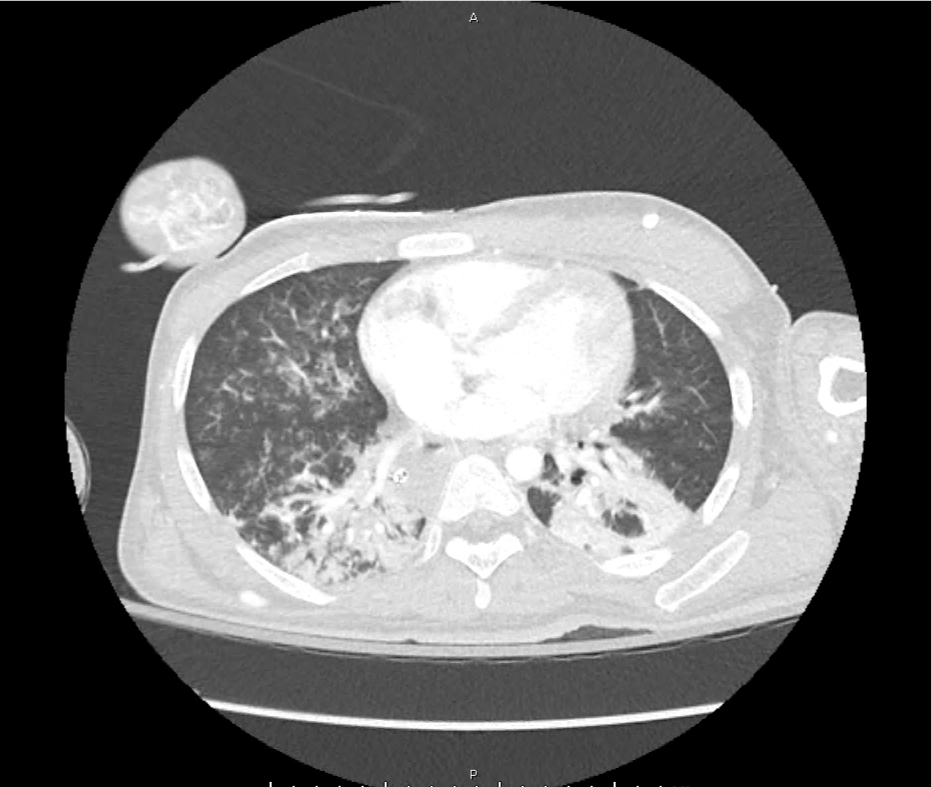
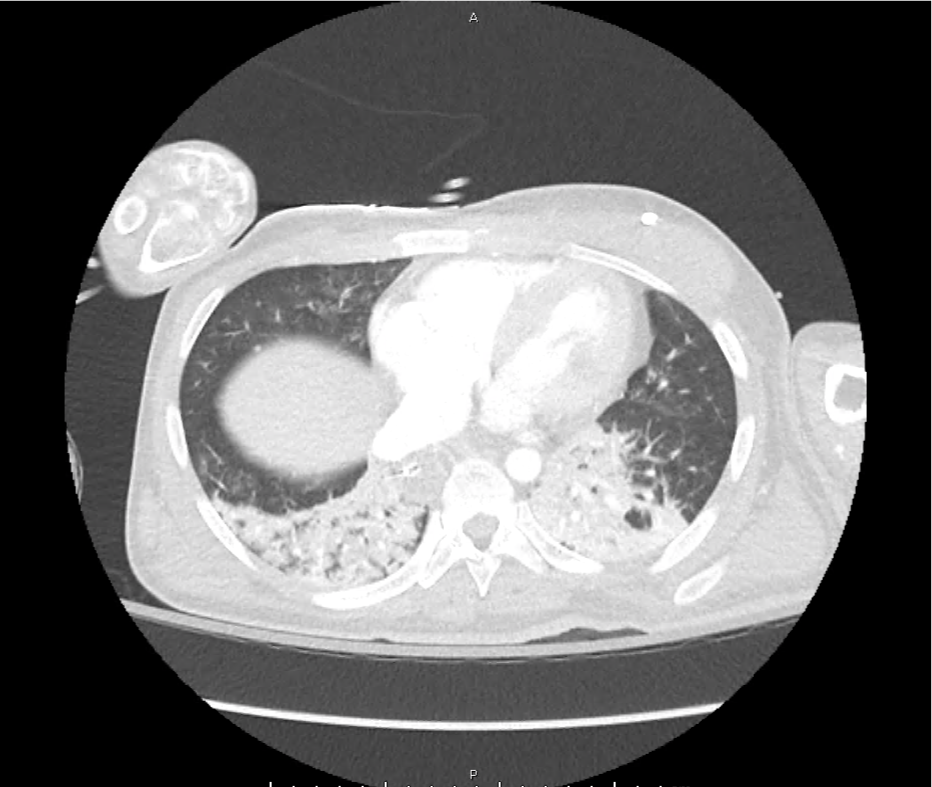
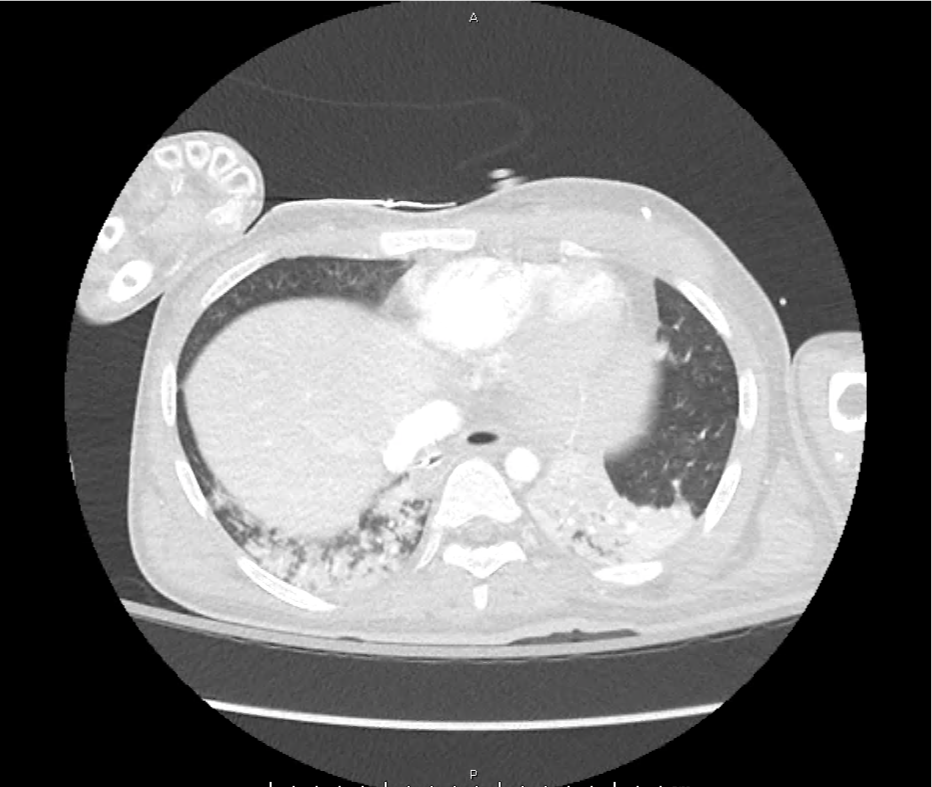
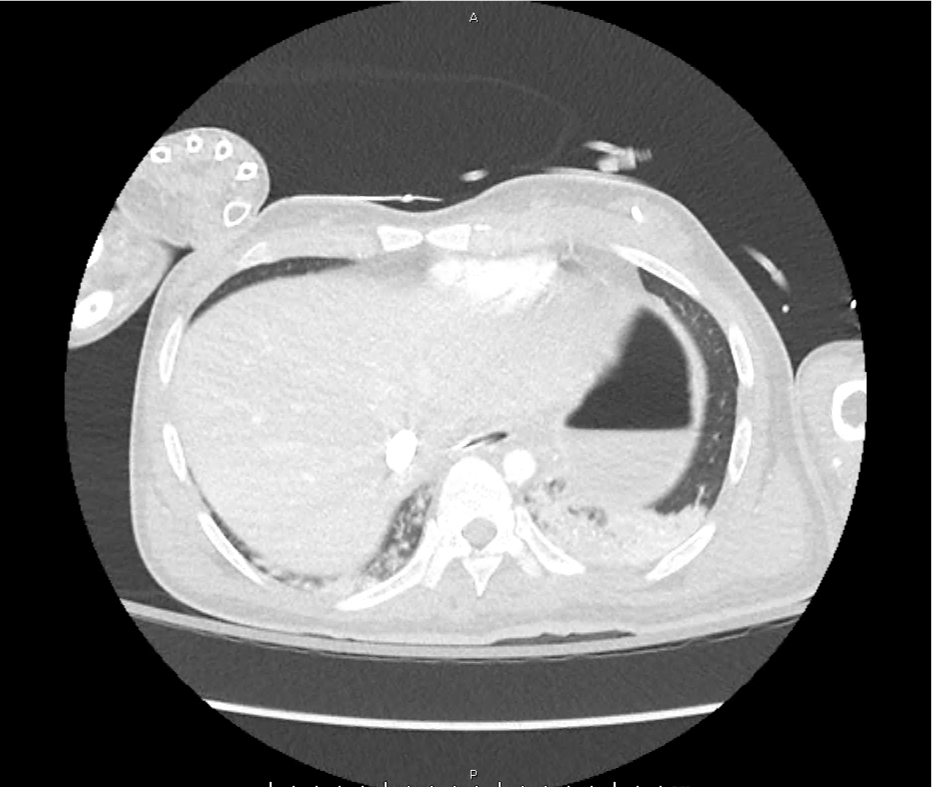
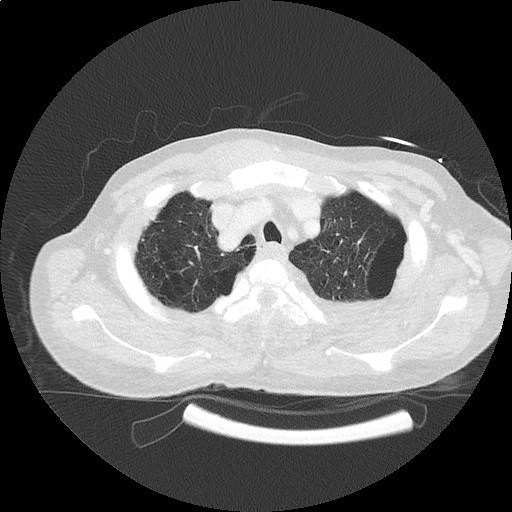
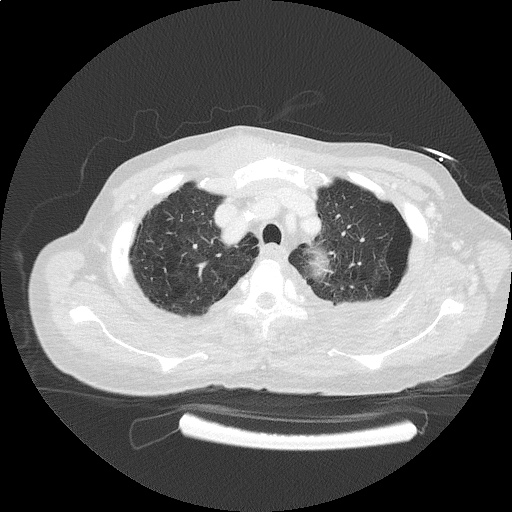
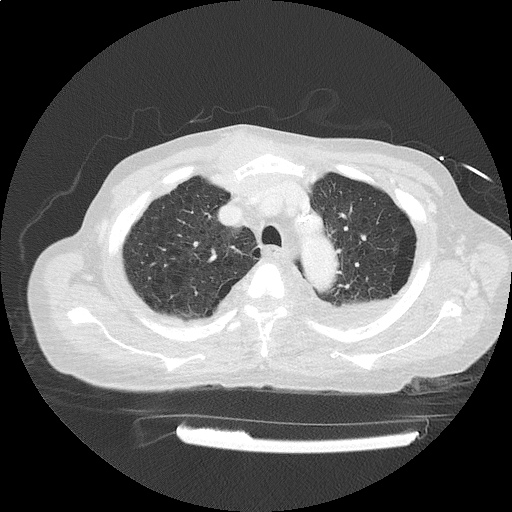
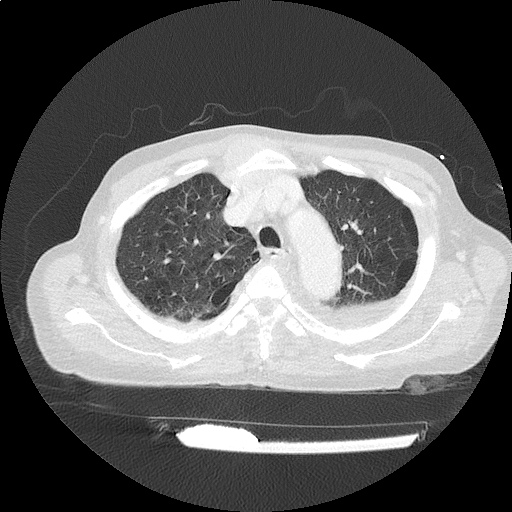
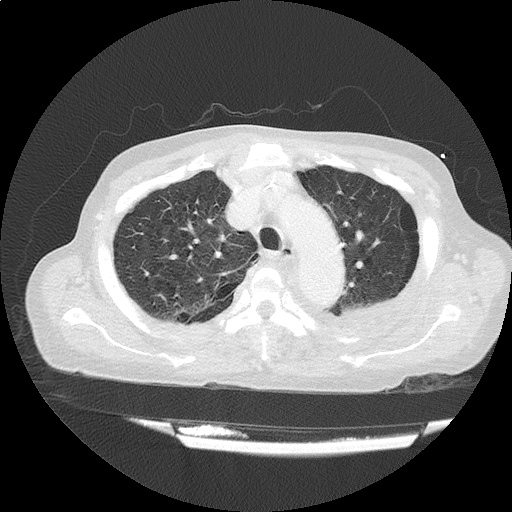
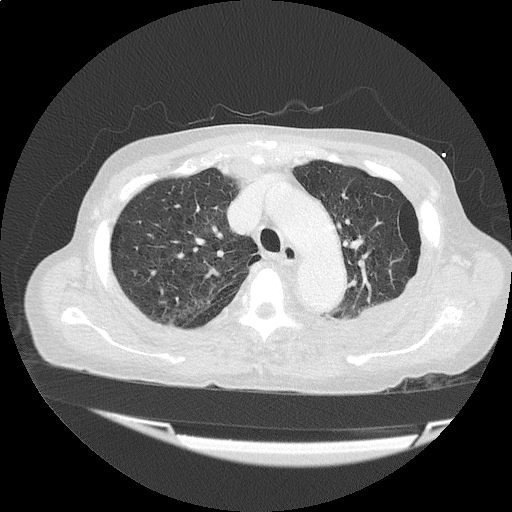
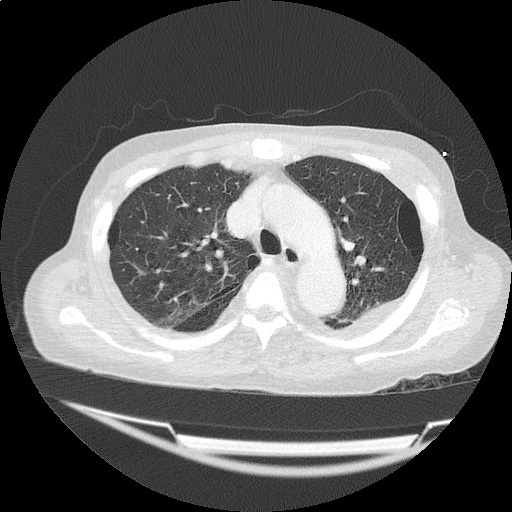
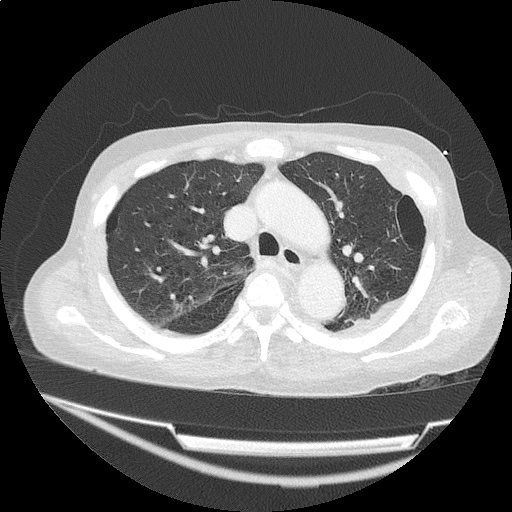
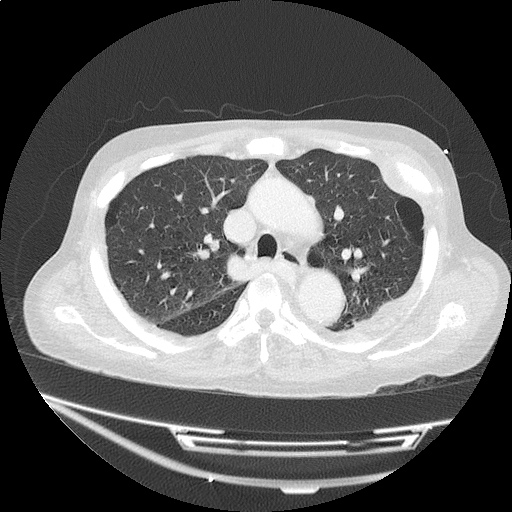
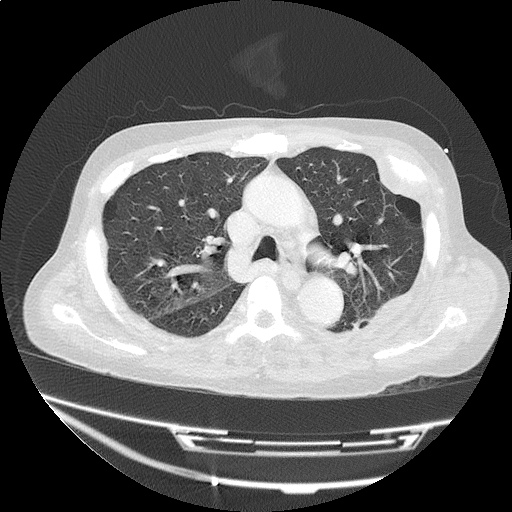
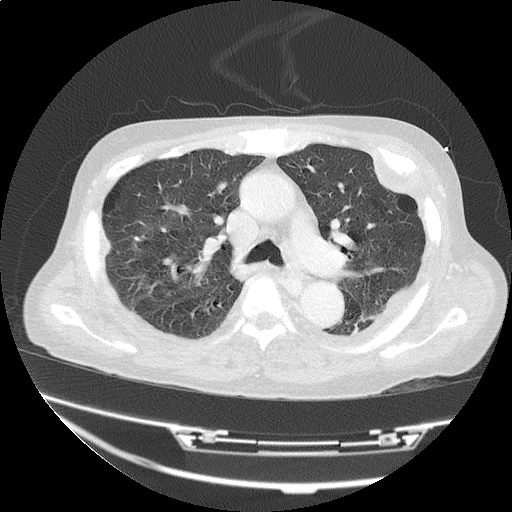
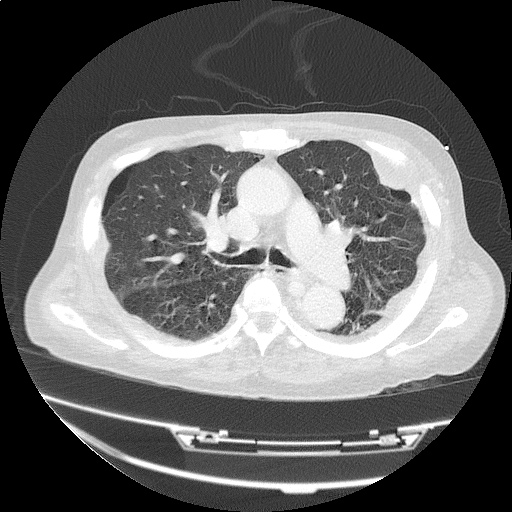
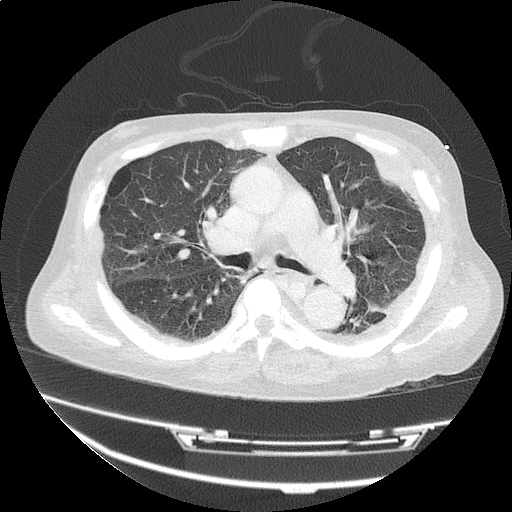
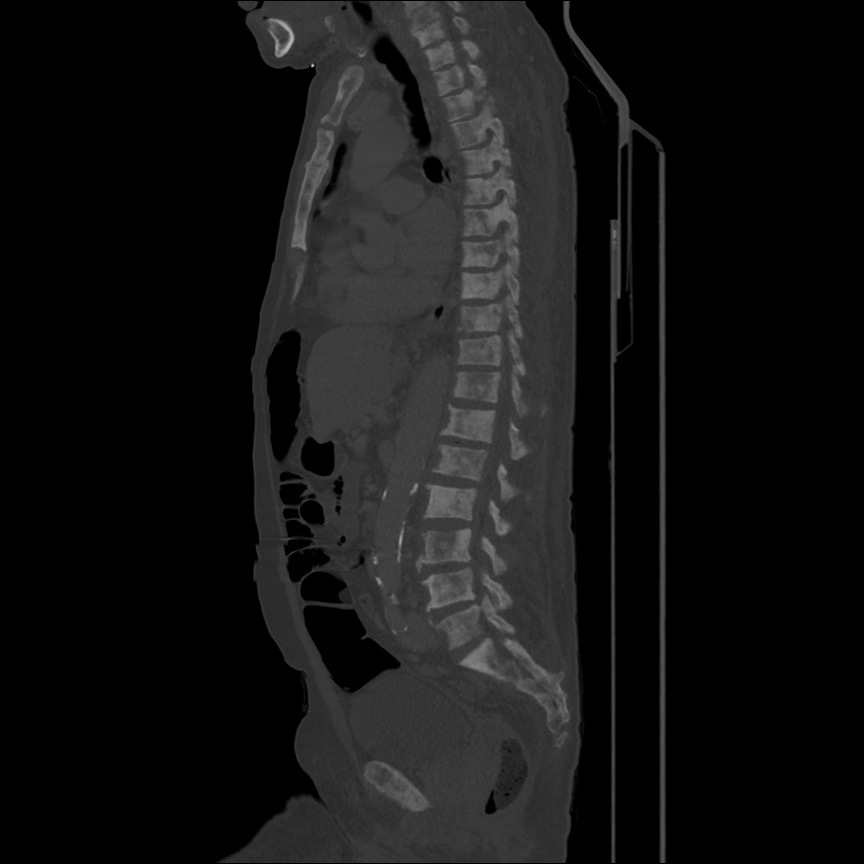
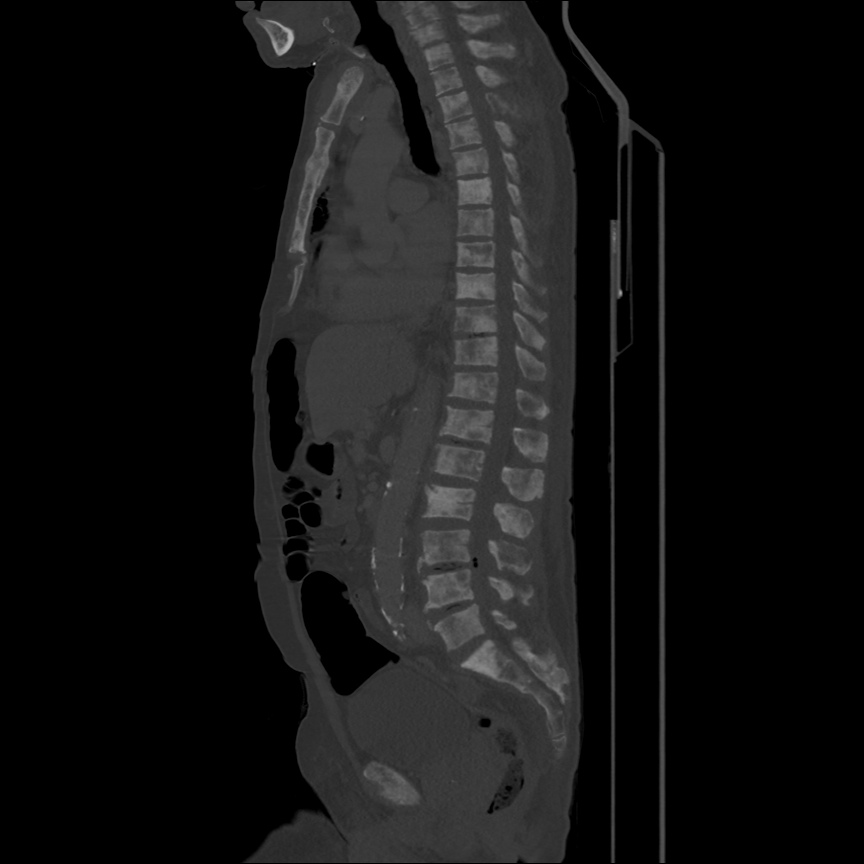
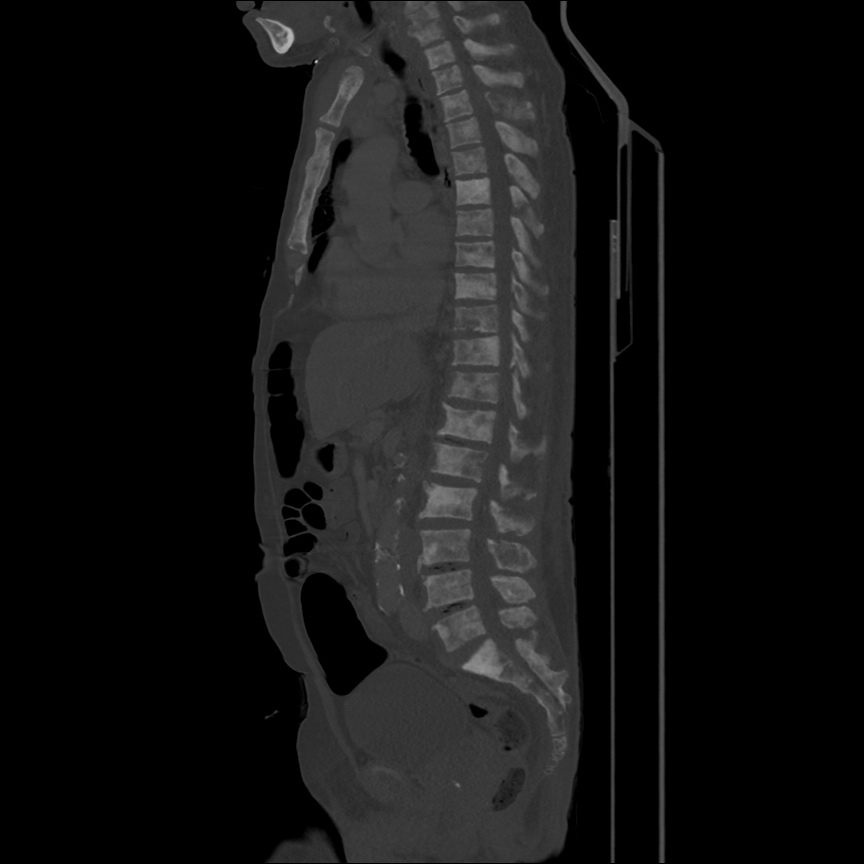
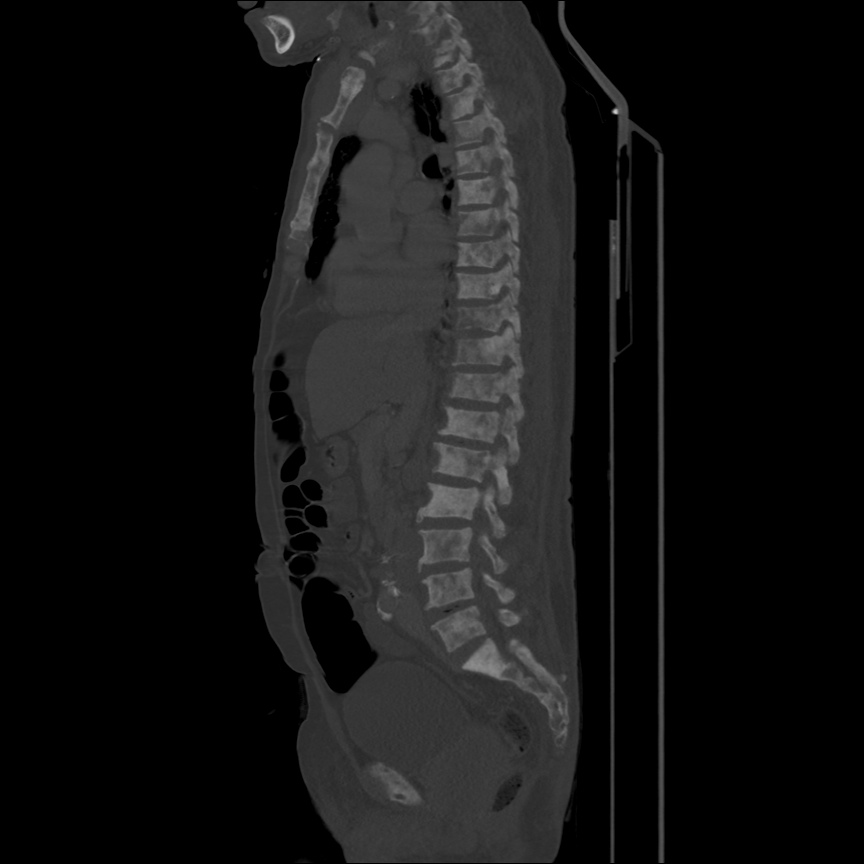
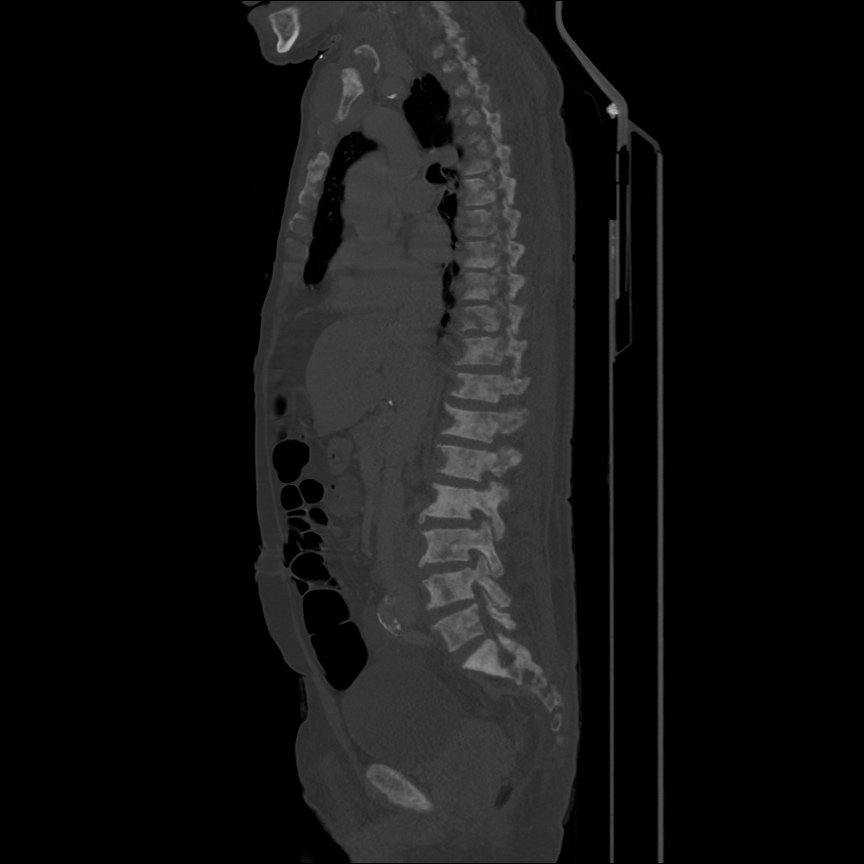
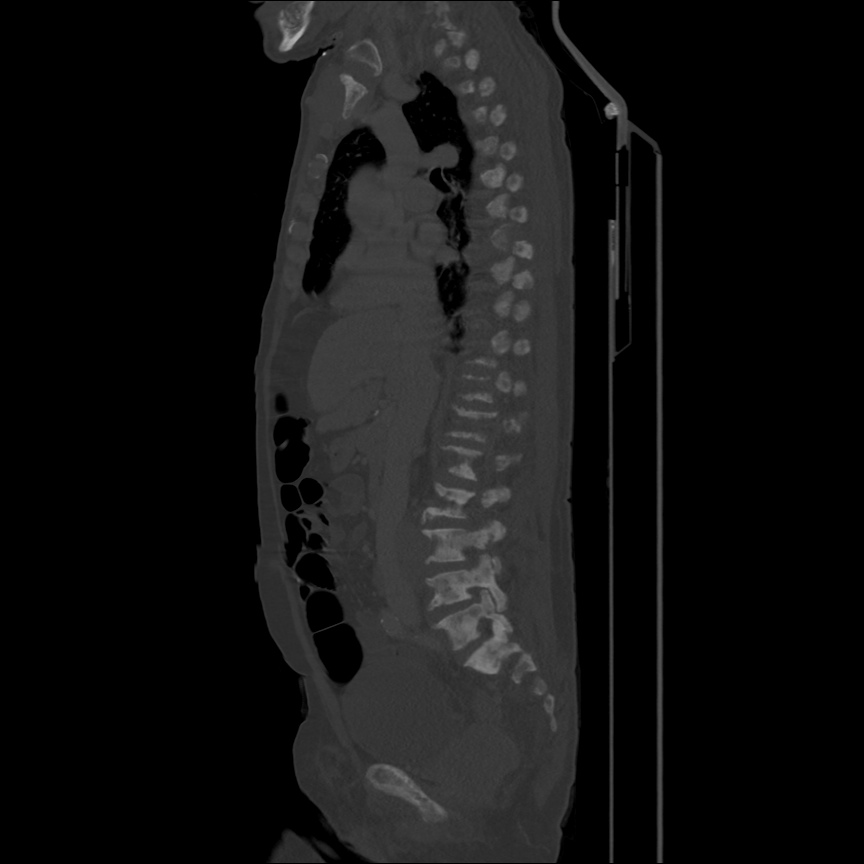
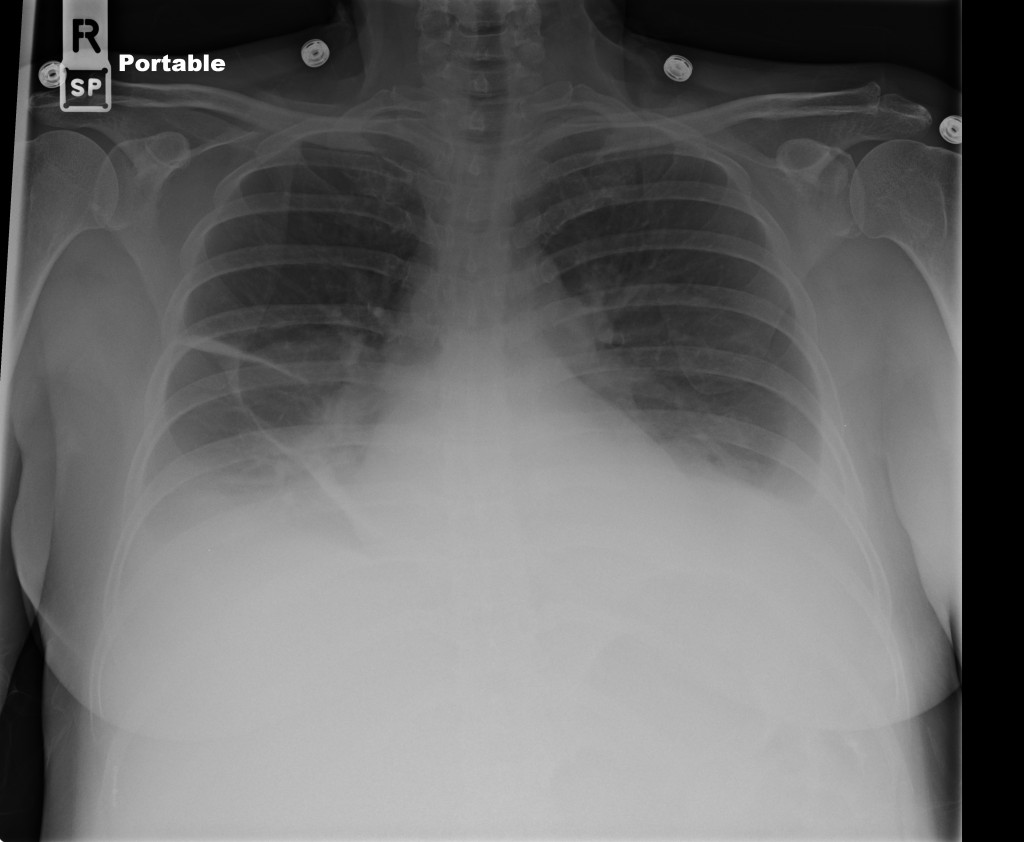
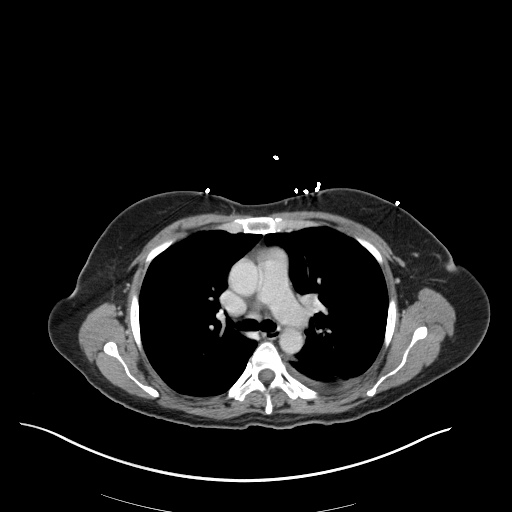
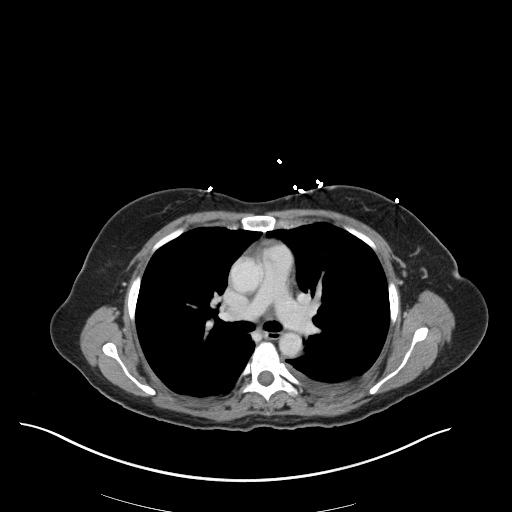
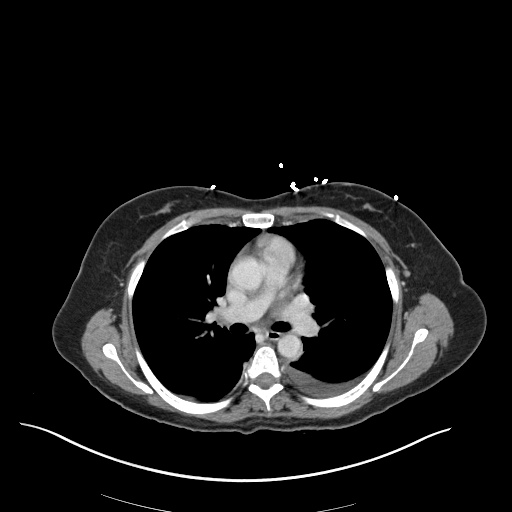
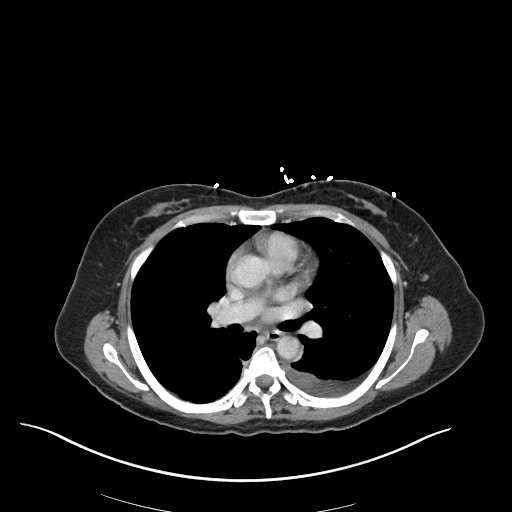
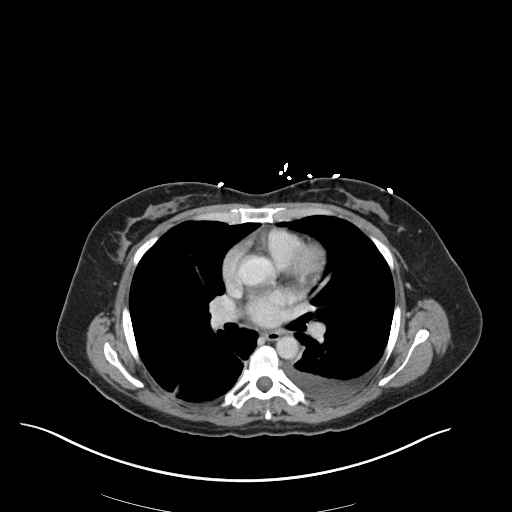
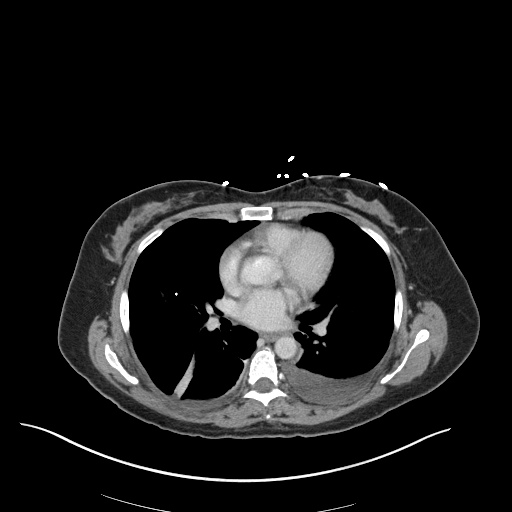
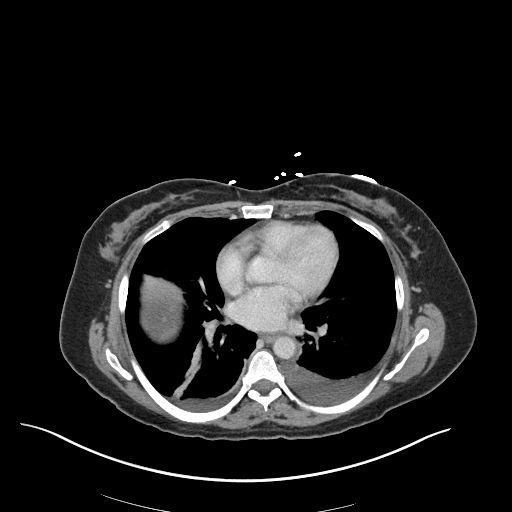
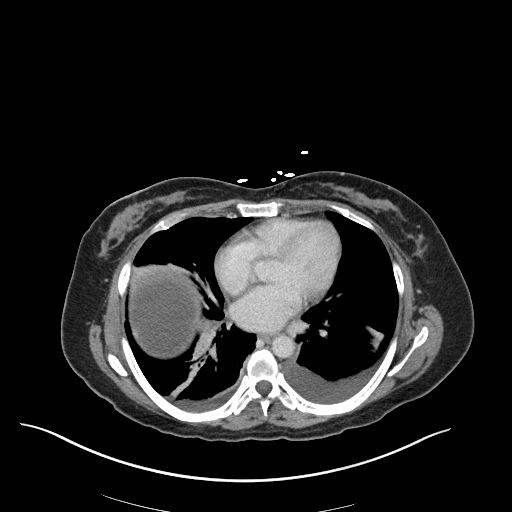
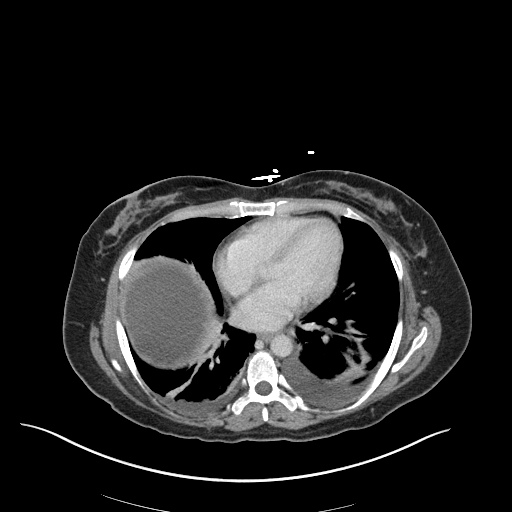
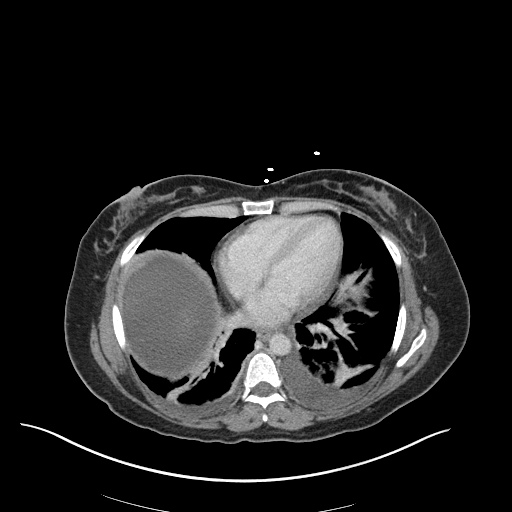
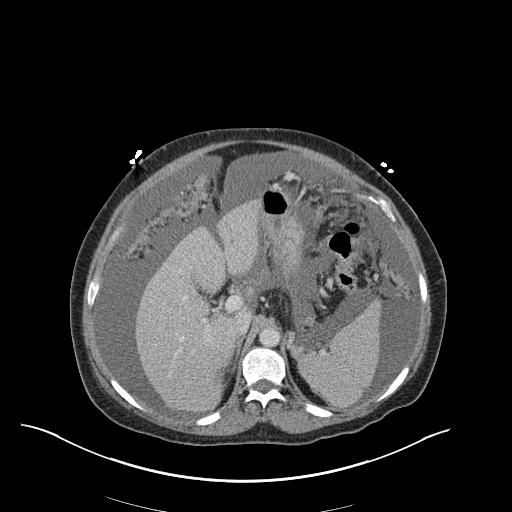
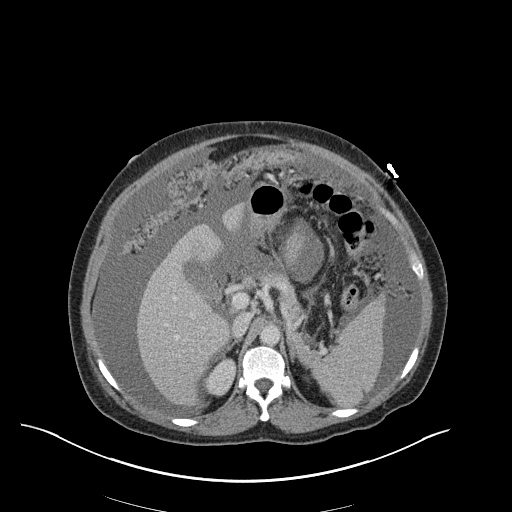
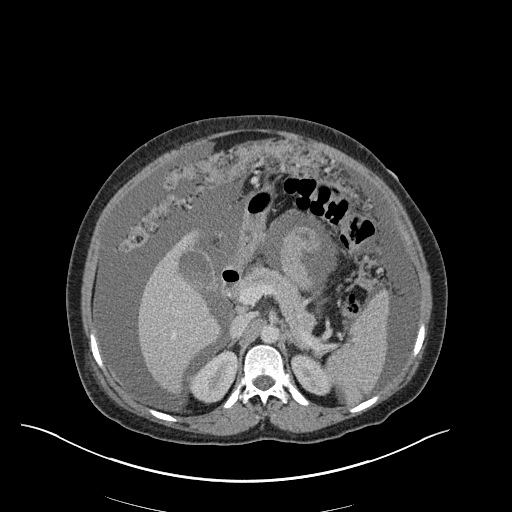
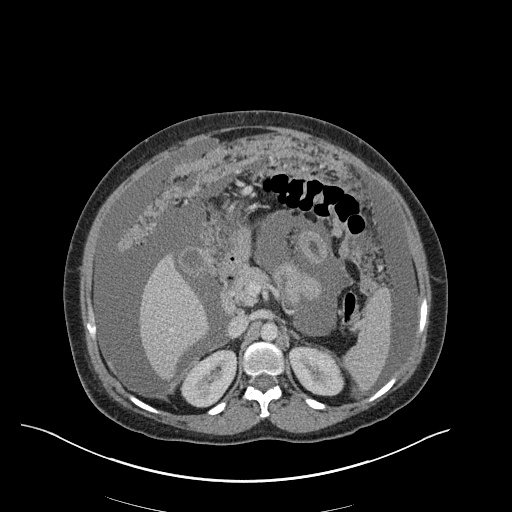
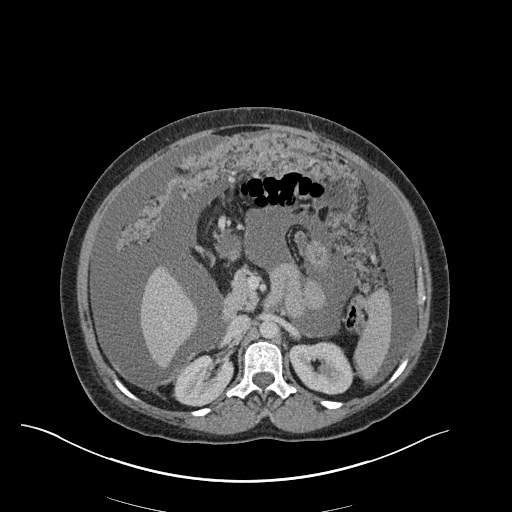
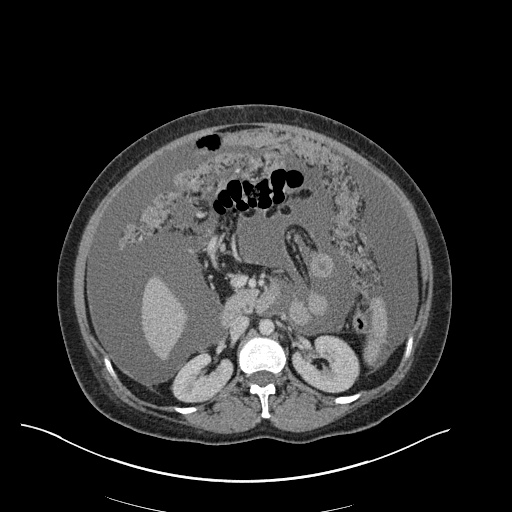
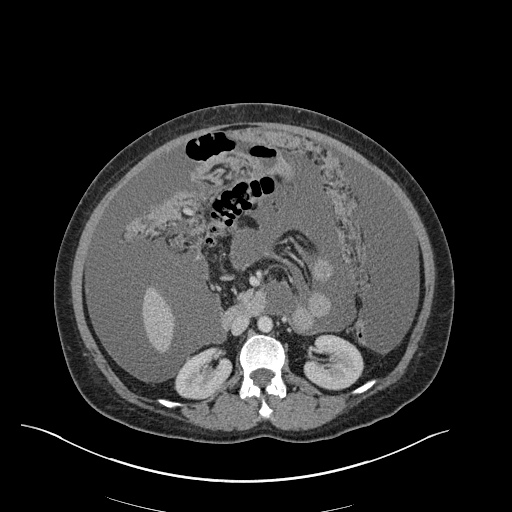
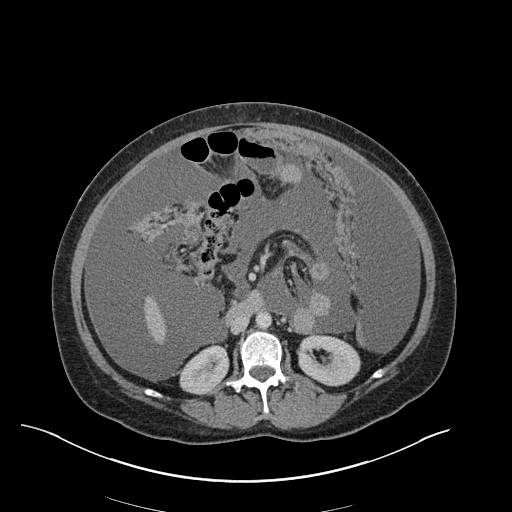
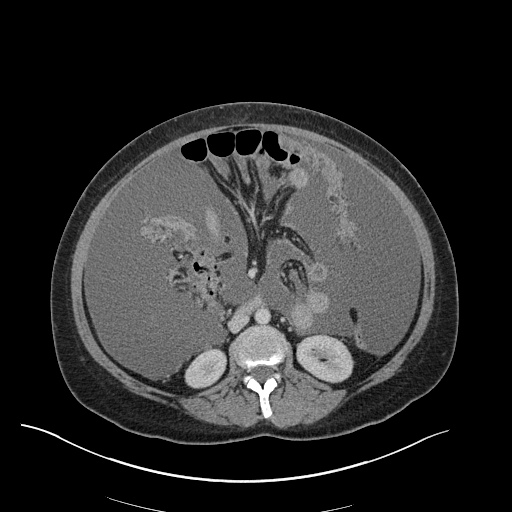
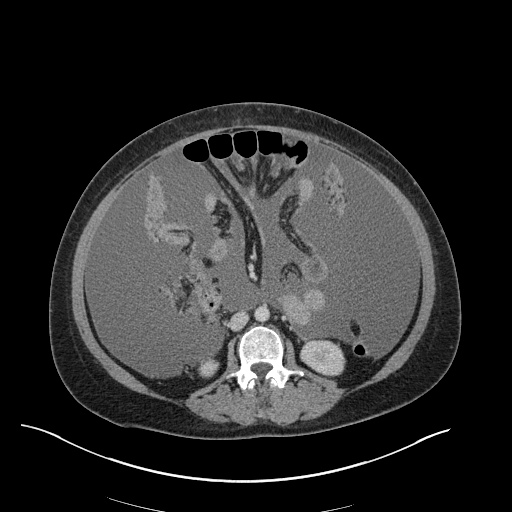
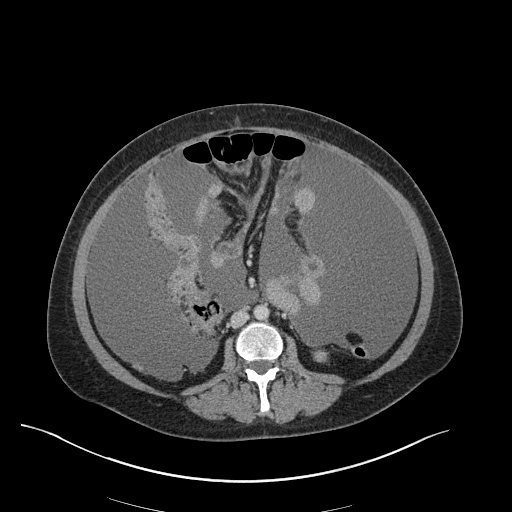
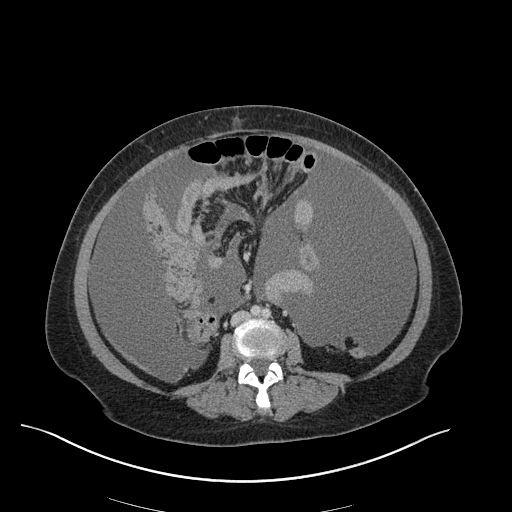
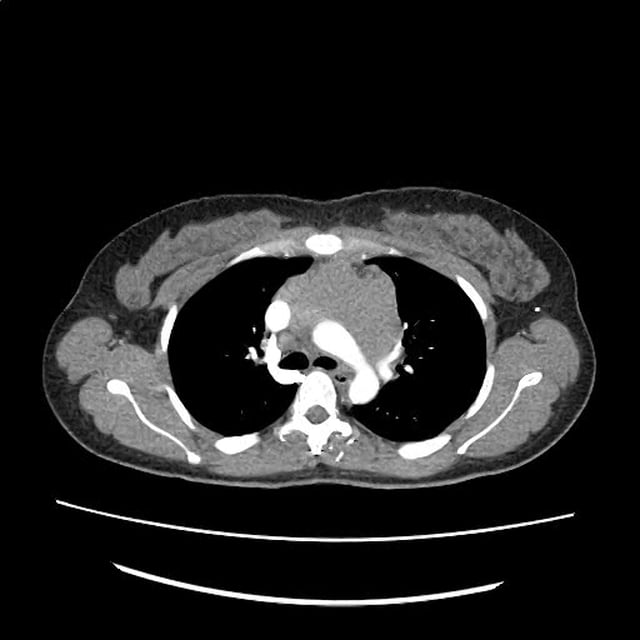
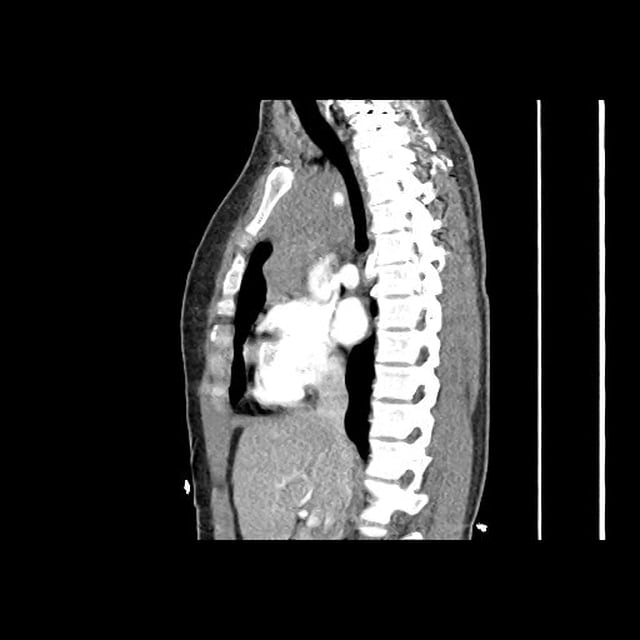
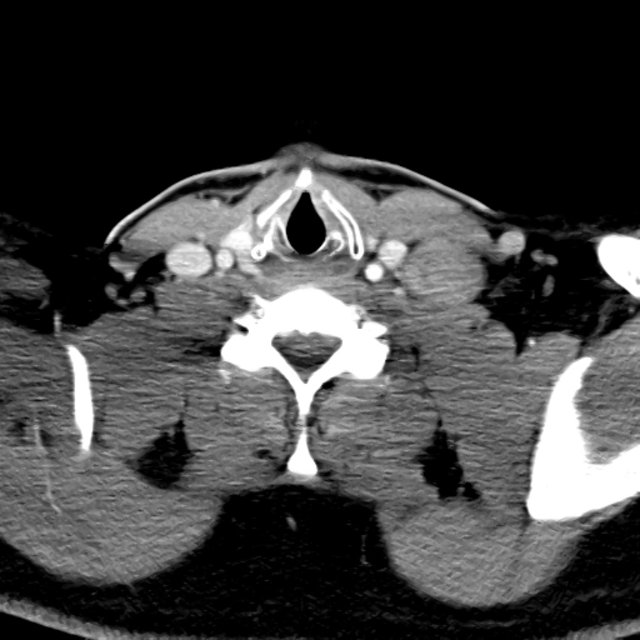
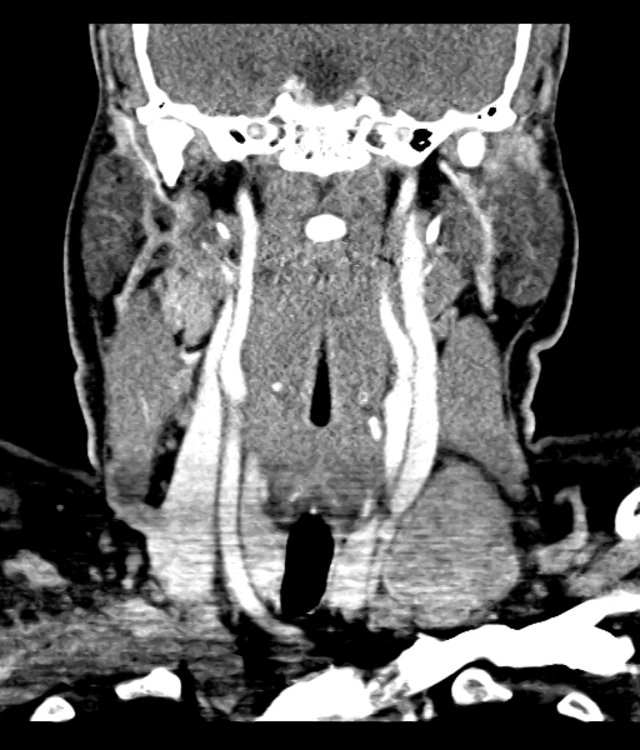
CT Pulmonary Angiography
Peribronchial opacities and patchy consolidation in the lungs which may represent multifocal pneumonia and/or aspiration in the appropriate clinical setting.
Mildly dilated main pulmonary artery suggestive of pulmonary arterial hypertension.
ED Course:
The patient was admitted to the medical intensive care unit for cardiopulmonary arrest presumed secondary to hypoxia and septic shock from healthcare-associated pneumonia or aspiration. The markedly elevated white blood cell count was attributed to a combination of infection and tissue ischemia from transient global hypoperfusion.
Definition: 1
- Markedly elevated leukocyte (particularly neutrophil) count without hematologic malignancy
- Cutoff is variable, 25-50k
Review of Available Literature
- Retrospective review of 135 patients with WBC >25k 2
- 48% infection
- 15% malignancy
- 9% hemorrhage
- 12% glucocorticoid or granulocyte colony stimulating therapy
- Retrospective review of 173 patients with WBC >30k 3
- 48% infection (7% C. difficile)
- 28% tissue ischemia
- 7% obstetric process (vaginal or cesarean delivery)
- 5% malignancy
- Observational study of 54 patients with WBC >25k 4
- Consecutive patients presenting to the emergency department
- Compared to age-matched controls with moderate leukocytosis (12-24k)
- Patients with leukemoid reaction were more likely to have an infection, be hospitalized and die.
Differential Diagnosis of Leukemoid Reaction 1,5-8
References
- Sakka V, Tsiodras S, Giamarellos-Bourboulis EJ, Giamarellou H. An update on the etiology and diagnostic evaluation of a leukemoid reaction. Eur J Intern Med. 2006;17(6):394-398. doi:10.1016/j.ejim.2006.04.004.
- Reding MT, Hibbs JR, Morrison VA, Swaim WR, Filice GA. Diagnosis and outcome of 100 consecutive patients with extreme granulocytic leukocytosis. Am J Med. 1998;104(1):12-16.
- Potasman I, Grupper M. Leukemoid reaction: spectrum and prognosis of 173 adult patients. Clin Infect Dis. 2013;57(11):e177-e181. doi:10.1093/cid/cit562.
- Lawrence YR, Raveh D, Rudensky B, Munter G. Extreme leukocytosis in the emergency department. QJM. 2007;100(4):217-223. doi:10.1093/qjmed/hcm006.
- Marinella MA, Burdette SD, Bedimo R, Markert RJ. Leukemoid reactions complicating colitis due to Clostridium difficile. South Med J. 2004;97(10):959-963. doi:10.1097/01.SMJ.0000054537.20978.D4.
- Okun DB, Tanaka KR. Profound leukemoid reaction in cytomegalovirus mononucleosis. JAMA. 1978;240(17):1888-1889.
- Halkes CJM, Dijstelbloem HM, Eelkman Rooda SJ, Kramer MHH. Extreme leucocytosis: not always leukaemia. Neth J Med. 2007;65(7):248-251.
- Granger JM, Kontoyiannis DP. Etiology and outcome of extreme leukocytosis in 758 nonhematologic cancer patients: a retrospective, single-institution study. Cancer. 2009;115(17):3919-3923. doi:10.1002/cncr.24480.