Brief Progress Note
Notified by nursing of abnormal vital signs, SpO2 91%. Briefly, this patient is a 52 year-old G1P1 with no prior medical history who is post-operative day three status post total abdominal hysterectomy, bilateral salpingoophorectomy as well as tumor debulking and staging for suspected primary ovarian adenocarcinoma based on peritoneal fluid cyctology.
On evaluation, the patient denied shortness of breath, chest pain, pleuritic chest pain, cough/hemoptysis, or calf/thigh pain. She states that she had been ambulating around the ward prior to having her vital signs assessed.
Physical Exam:
| VS: | T | 98.9 | HR | 94 | RR | 18 | BP | 117/72 | O2 | 91% RA |
| Gen: | No acute distress, speaking in full sentences. | |||||||||
| HEENT: | No jugular venous distension. | |||||||||
| CV: | RRR, normal S1/S2, no prominent P2, no additional heart sounds. | |||||||||
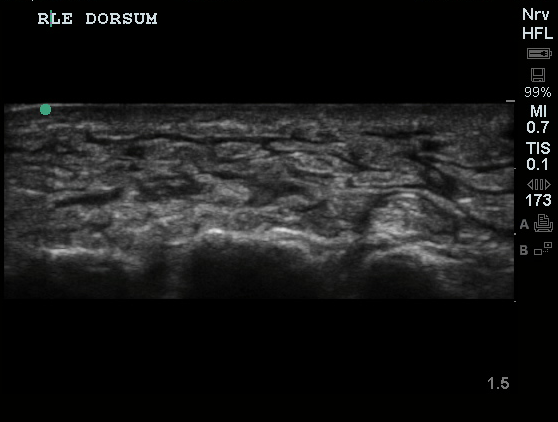
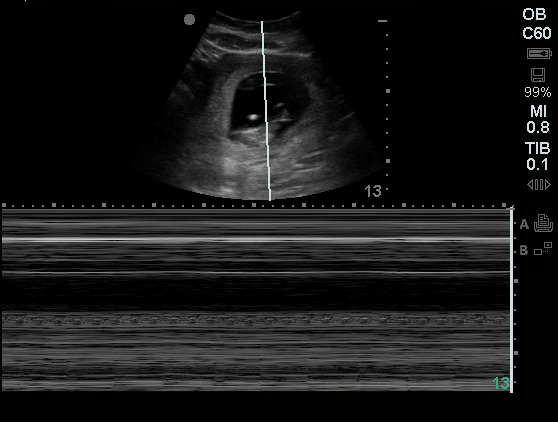
| Lungs: | Decreased breath sounds at inferior 1/3 posterior lung fields bilaterally, faint crackles above, no wheezing. Dullness to percussion in inferior lung fields. | |||||||||
| Ext: | Warm, well-perfused. Sequential compression devices on bilateral lower extremities, removed revealing trace pitting edema symmetric bilaterally, no tenderness to palpation of posterior leg, no pain with passive dorsiflexion. | |||||||||
Medications:
- Lovenox 40mg s.q. daily
- Norco 5-325mg p.o. q.4.h. p.r.n. pain
- Morphine 2mg i.v. q.3.h. p.r.n. breakthrough pain
- Colace 100mg p.o. b.i.d. p.r.n. constipation
- Zofran 4mg i.v. q.6.h. p.r.n. nausea/vomiting
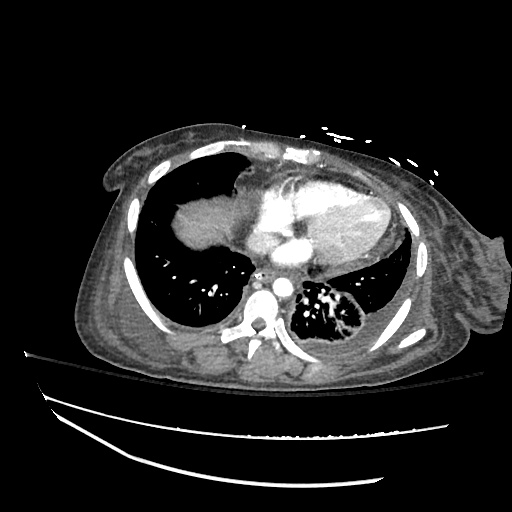
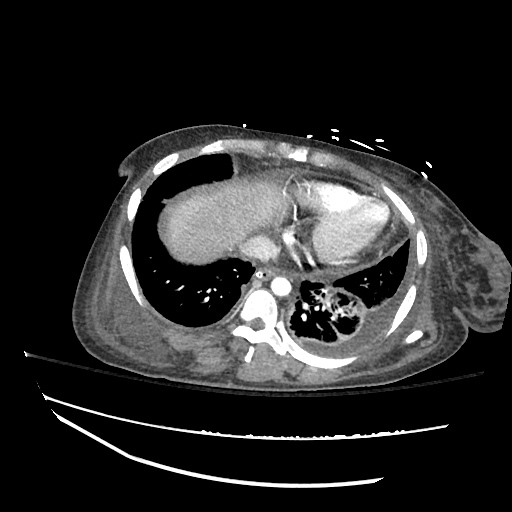
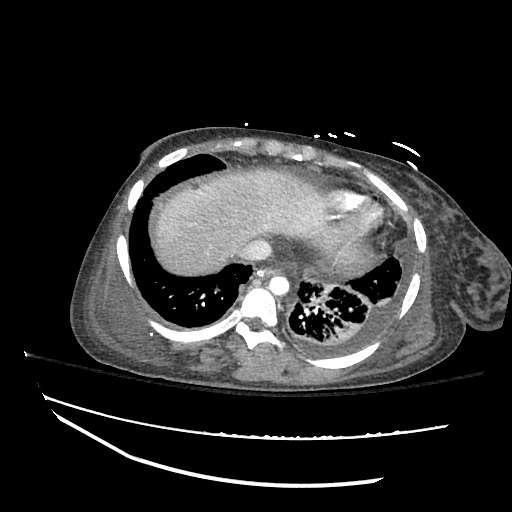
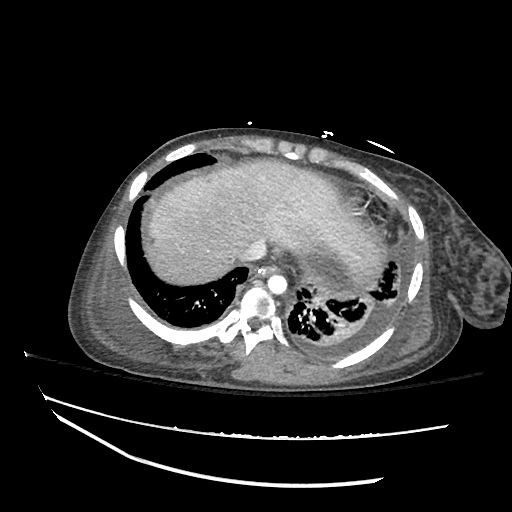
Imaging:
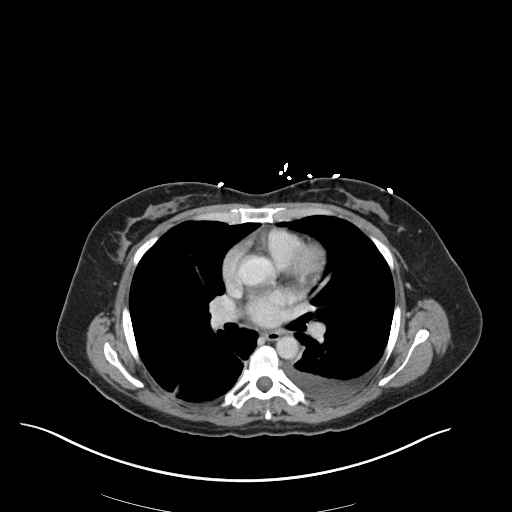
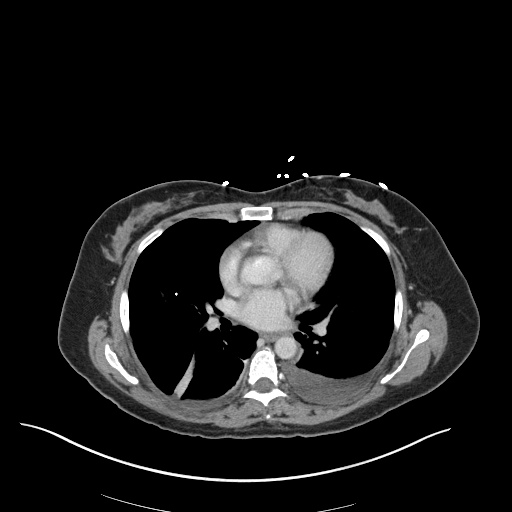
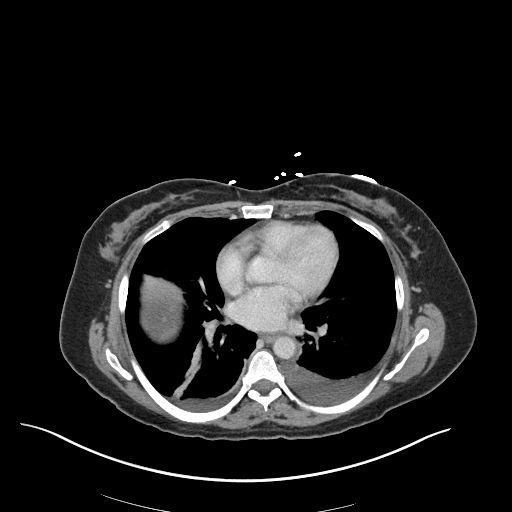
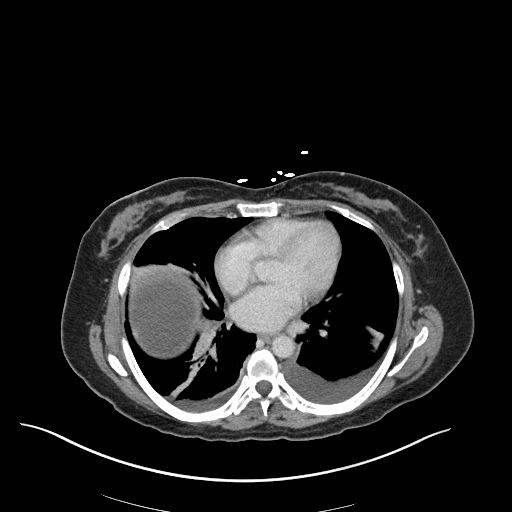
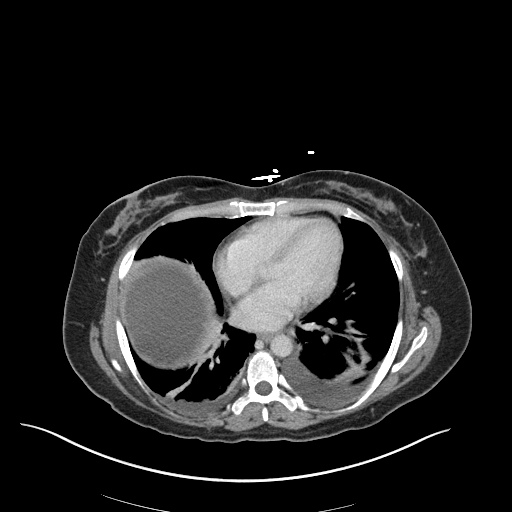
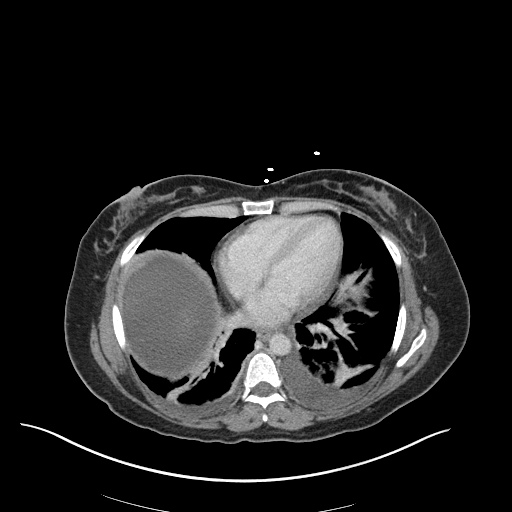
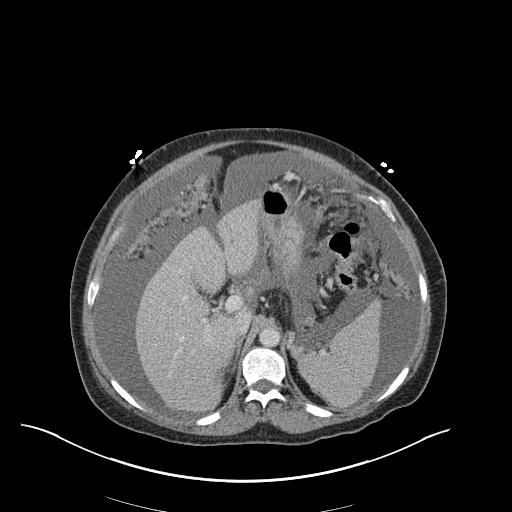
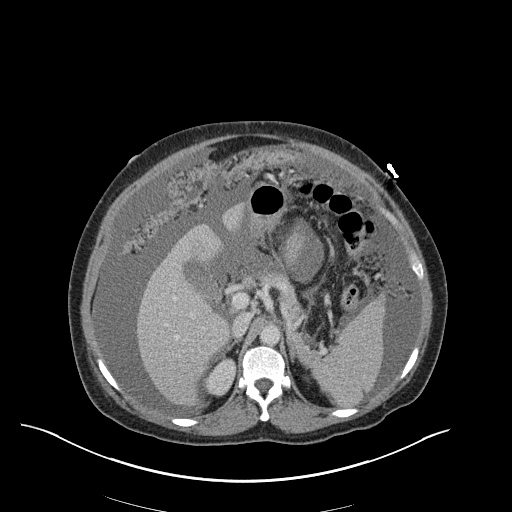
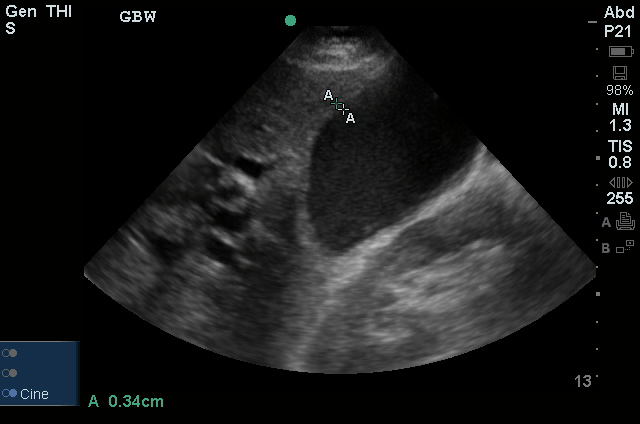
CXR
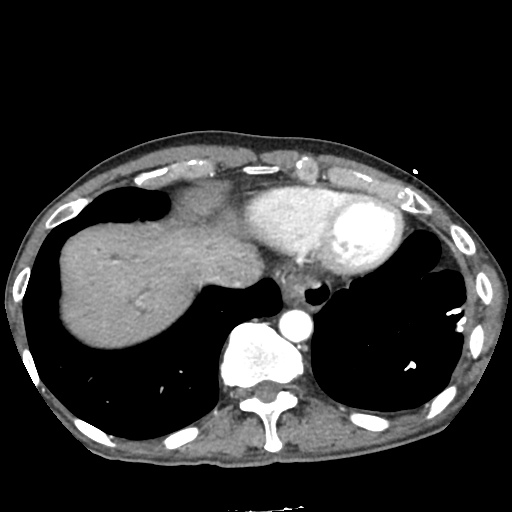
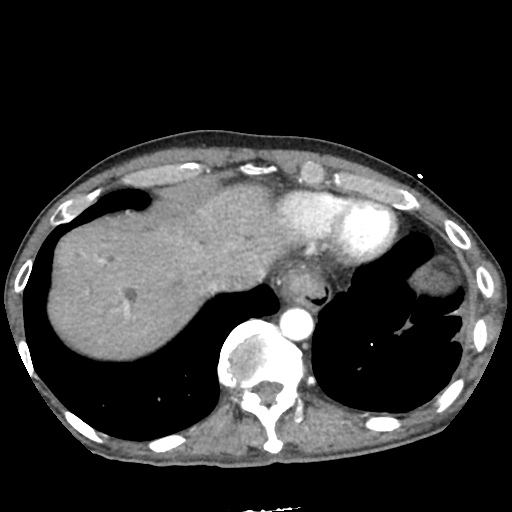
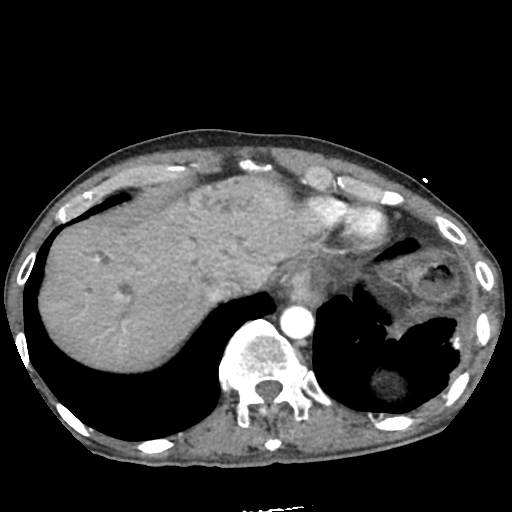
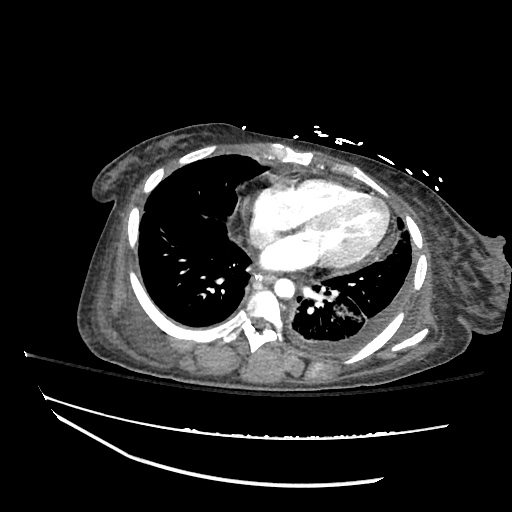
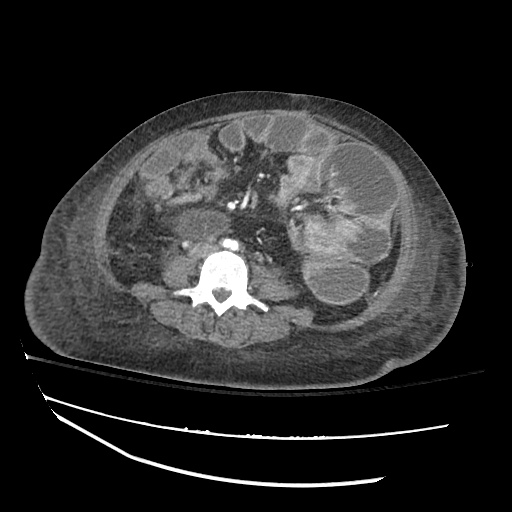
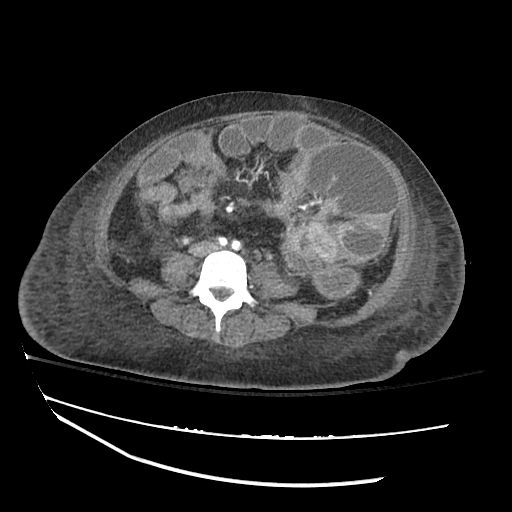
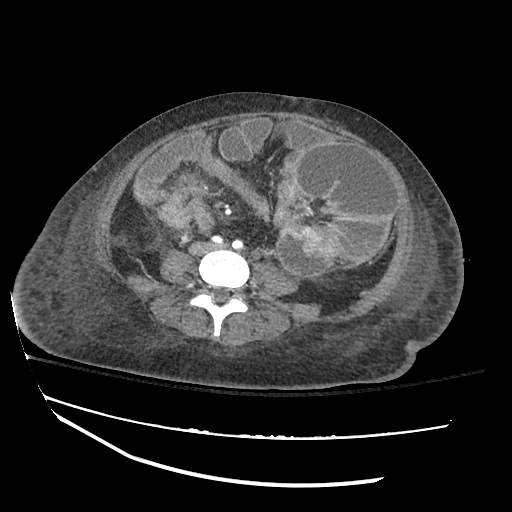
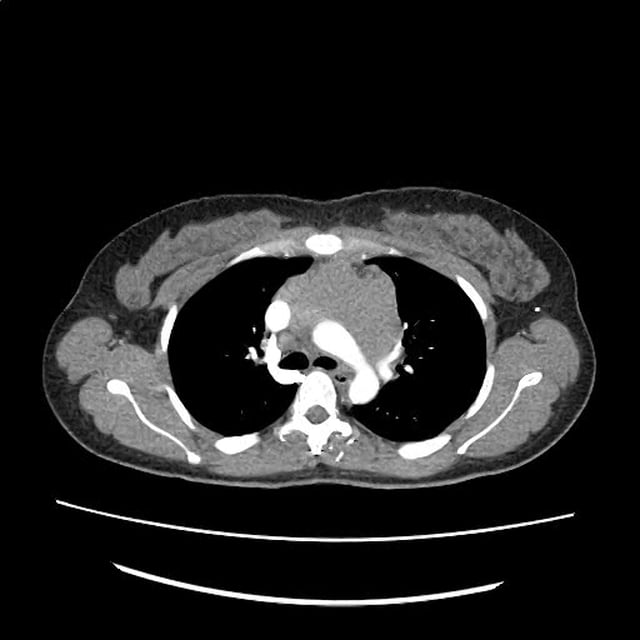
CT Chest
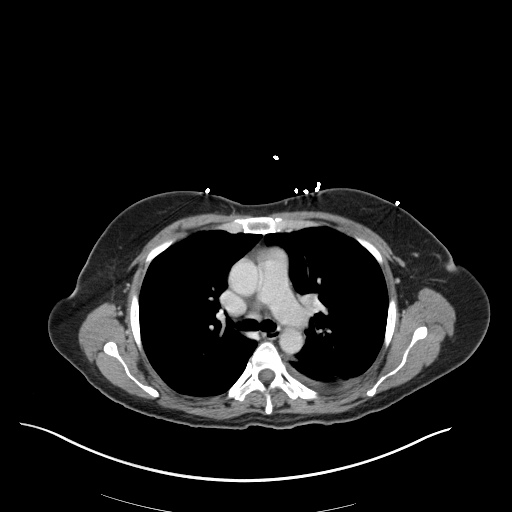
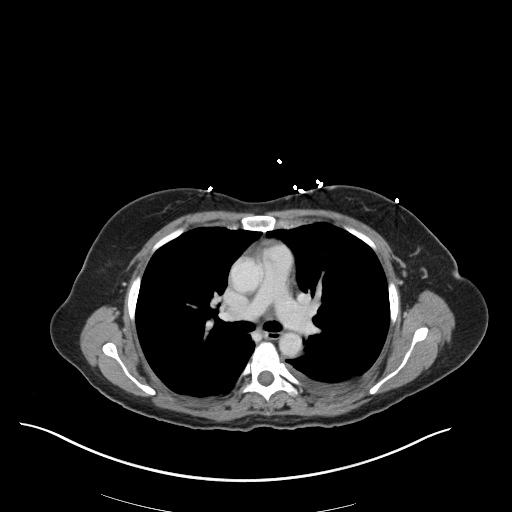
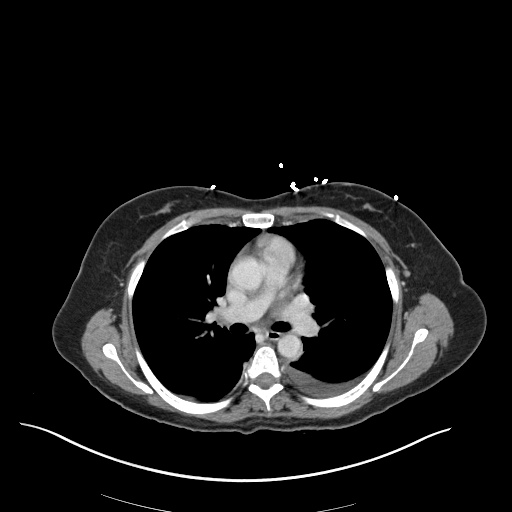
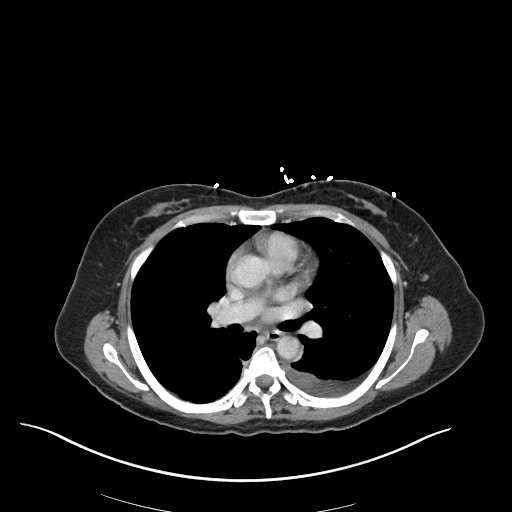
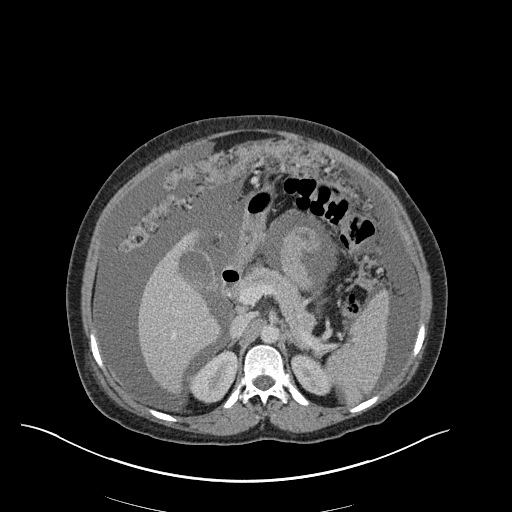
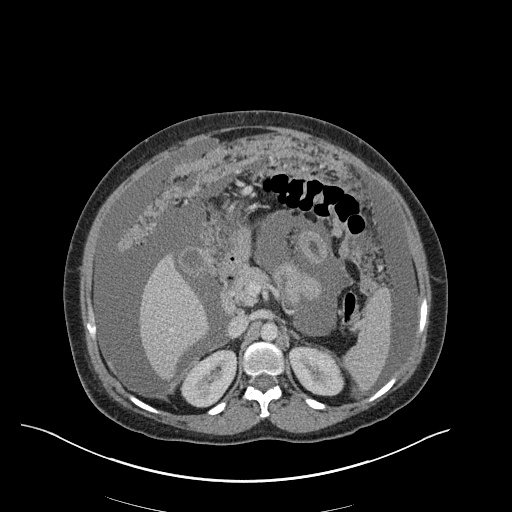
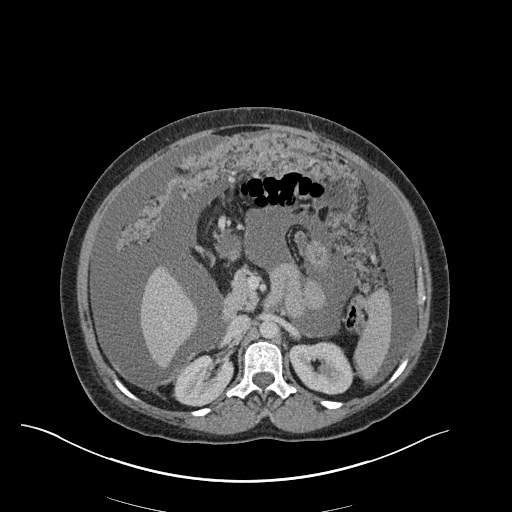
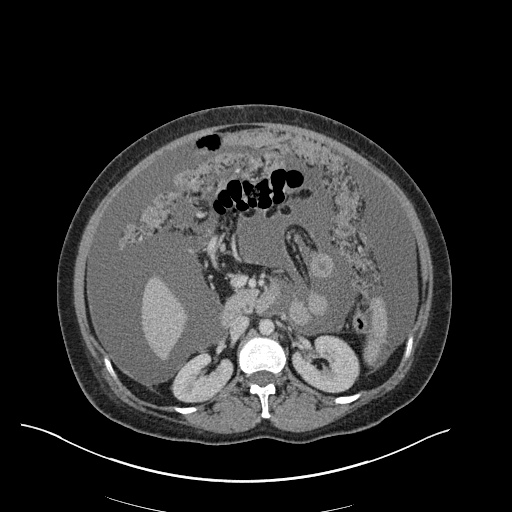
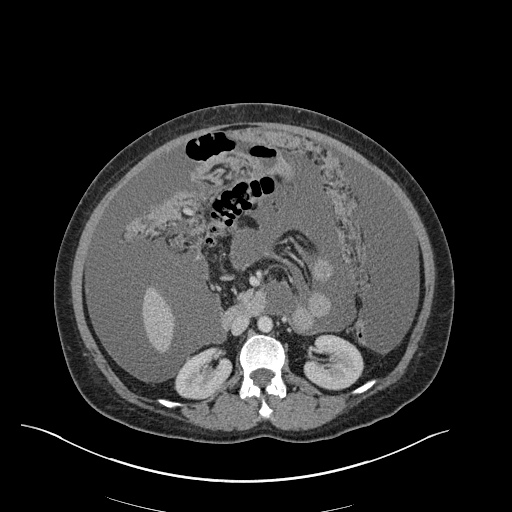
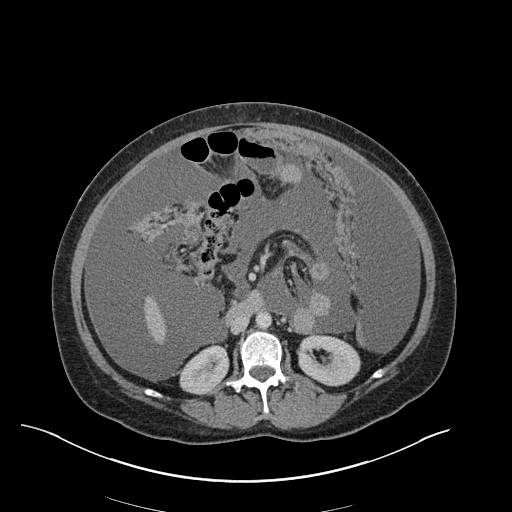
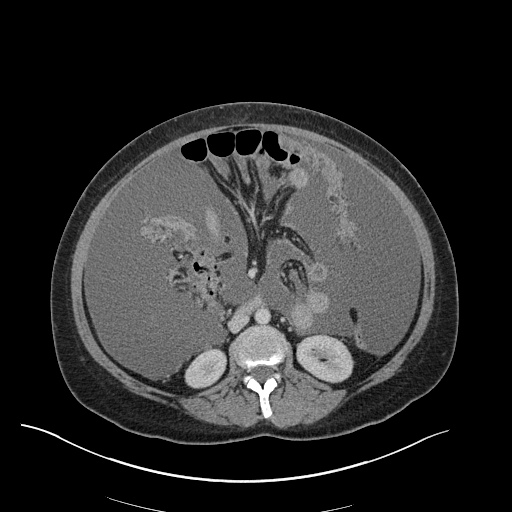
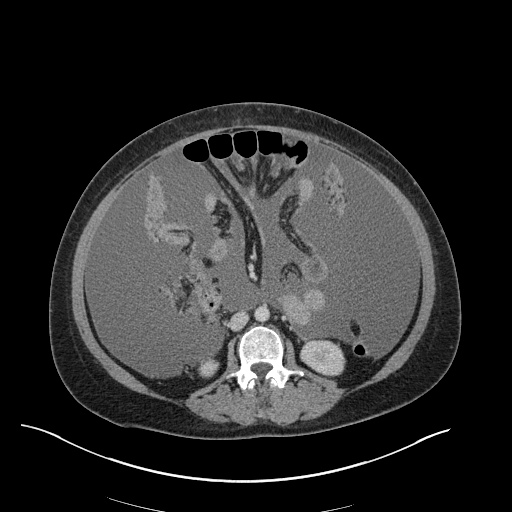
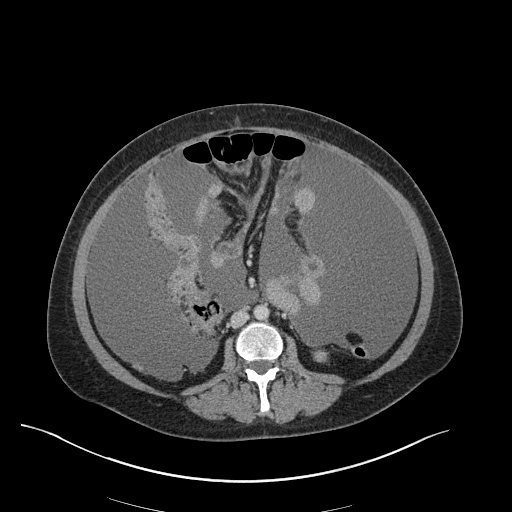
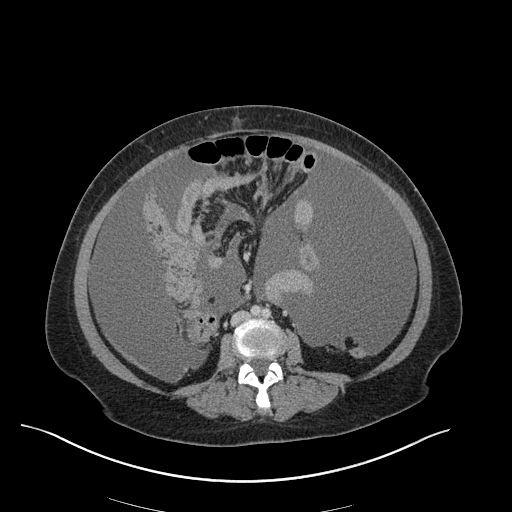
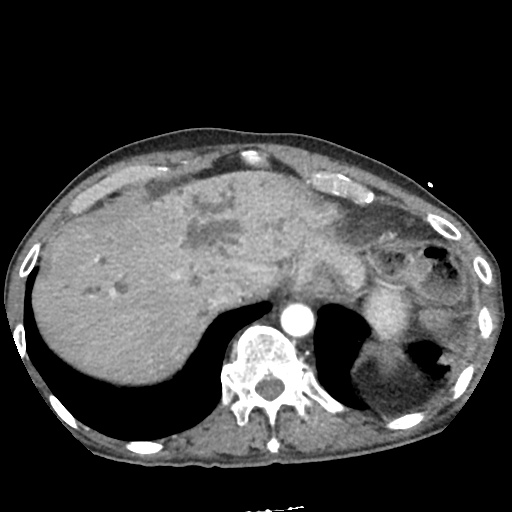
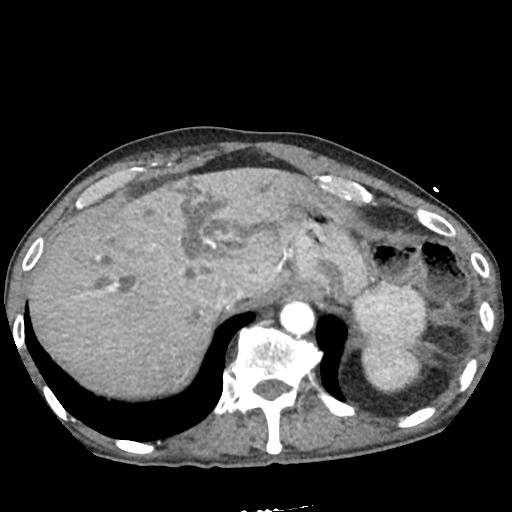
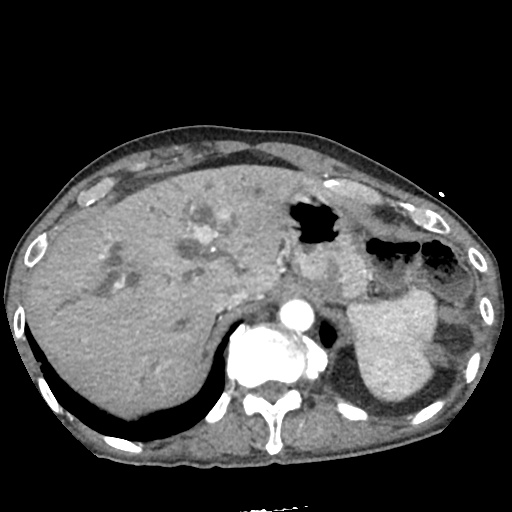
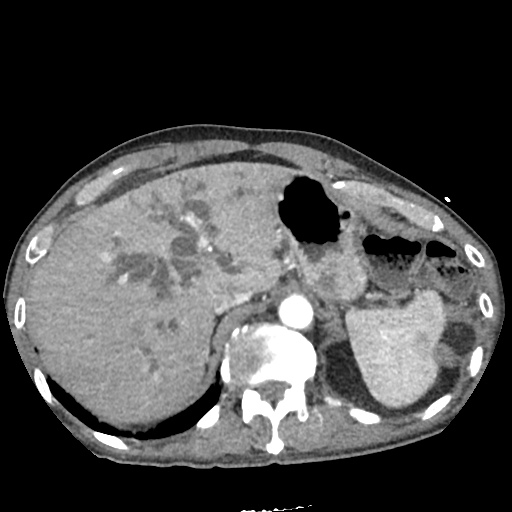
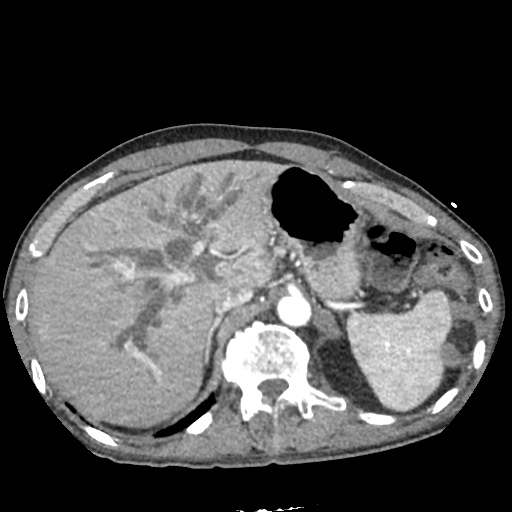
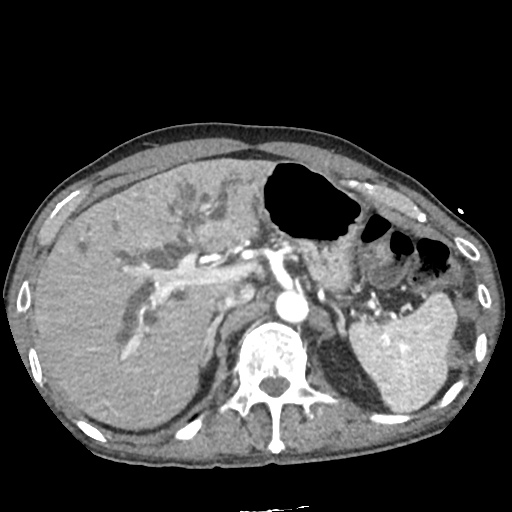
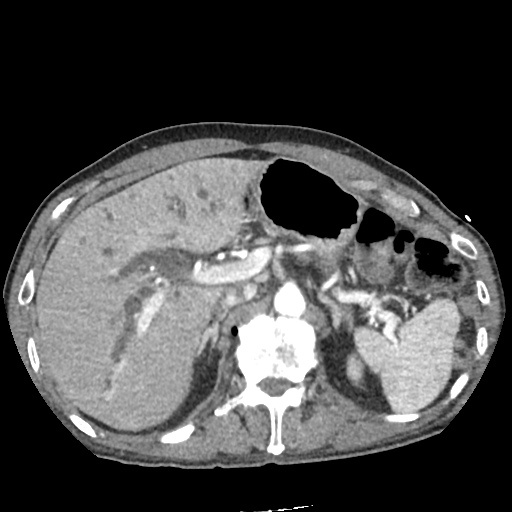
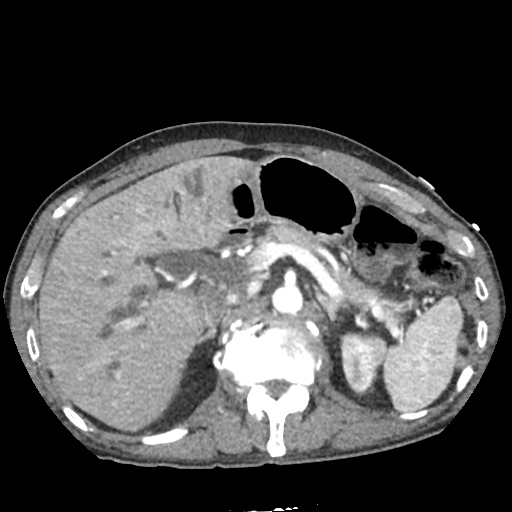
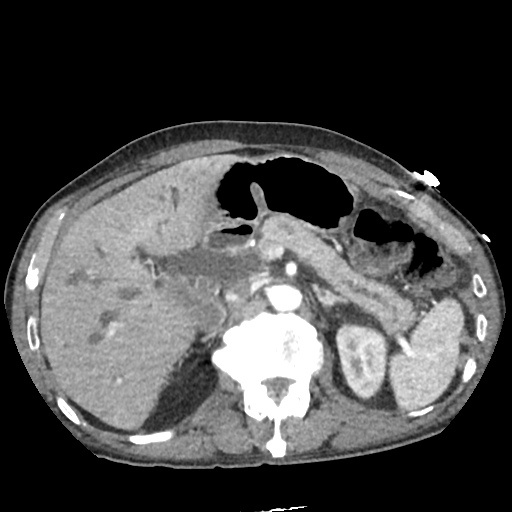
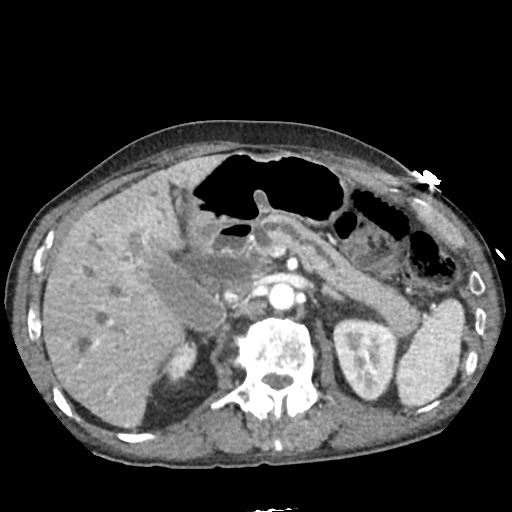
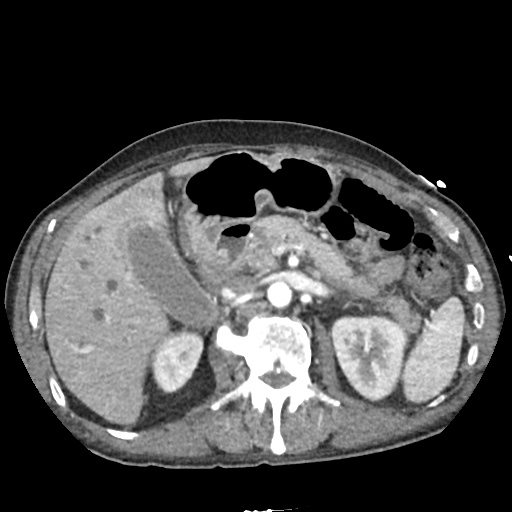
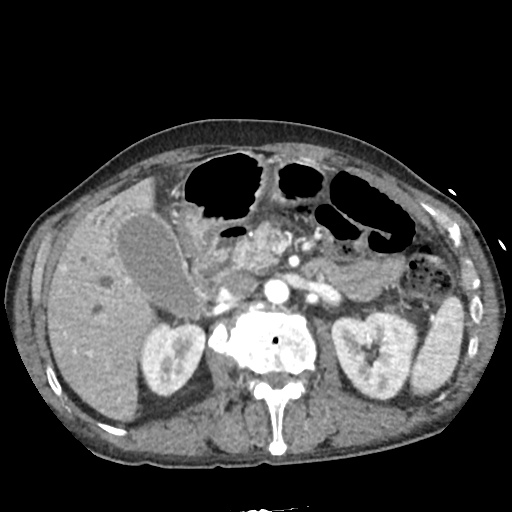
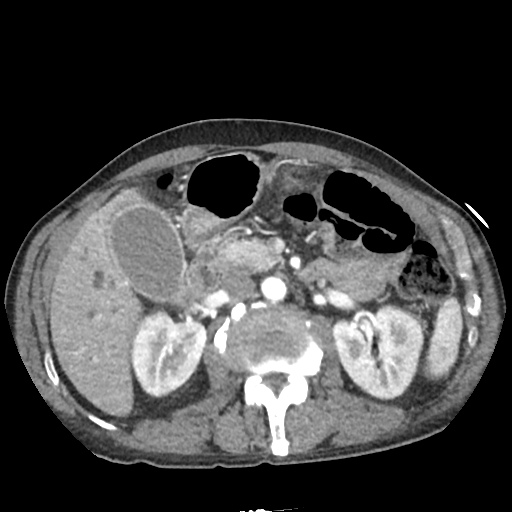
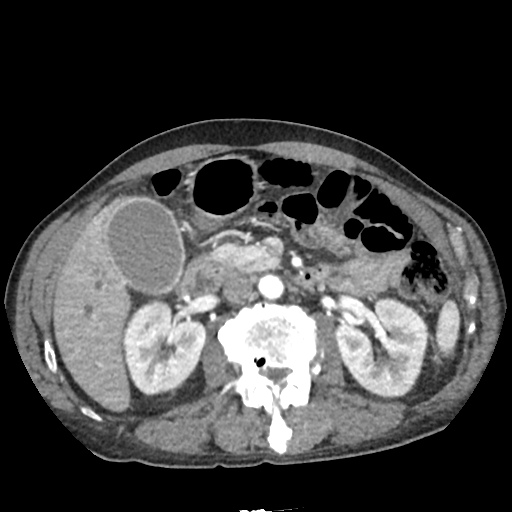
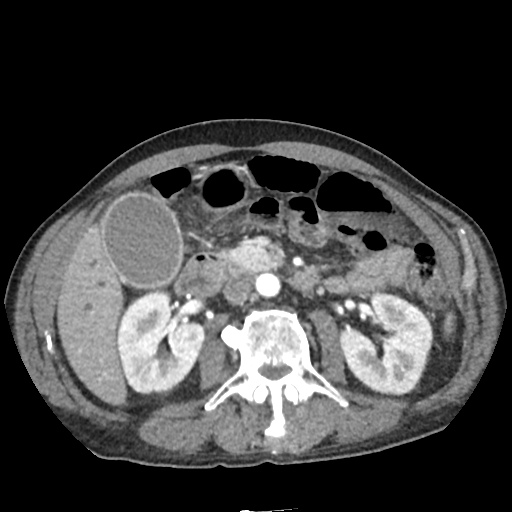
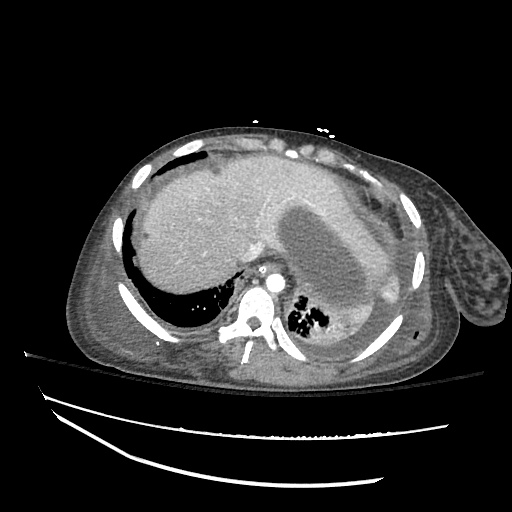
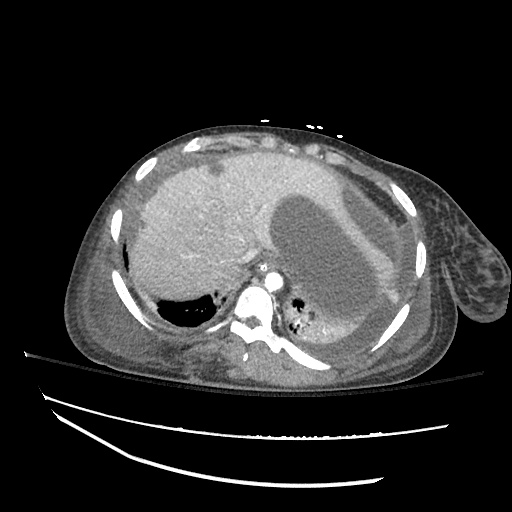
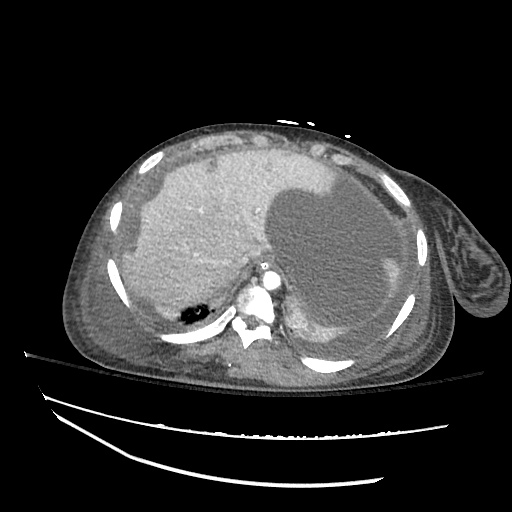
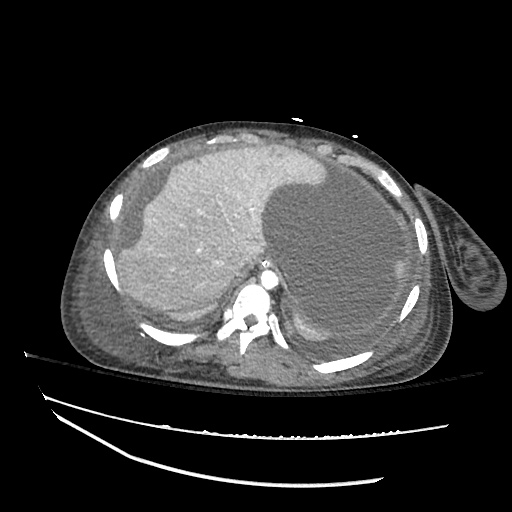
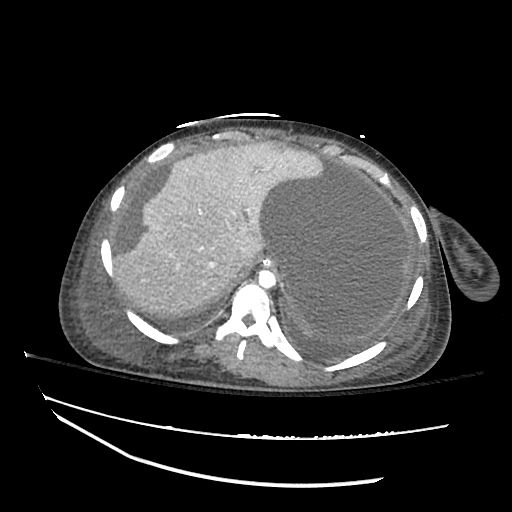
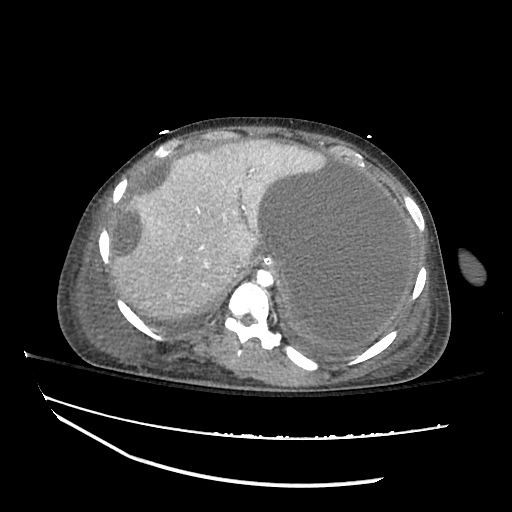
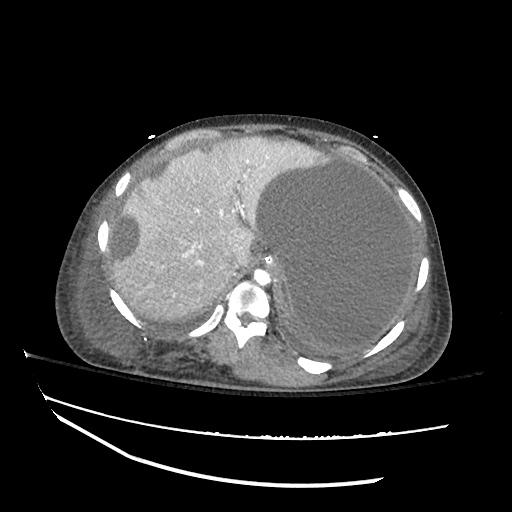
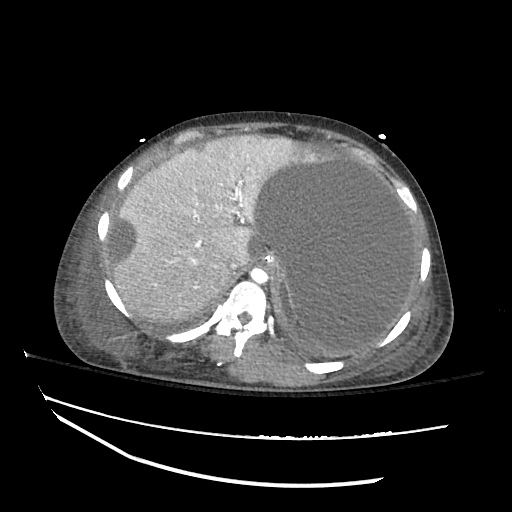
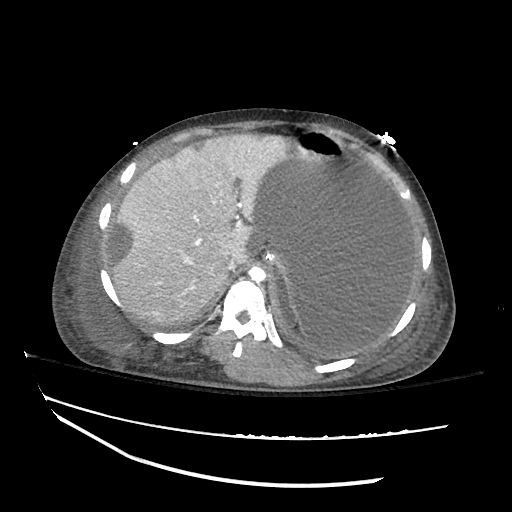
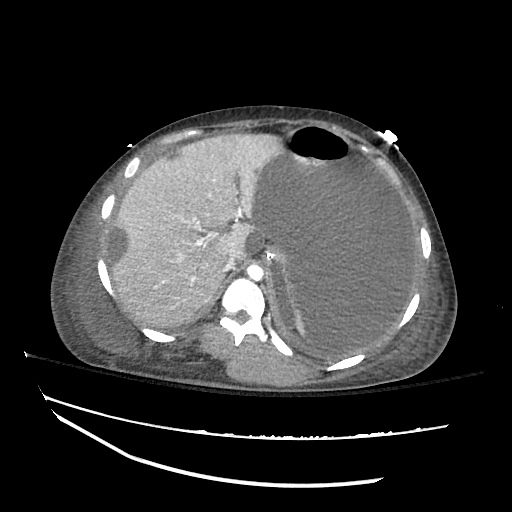
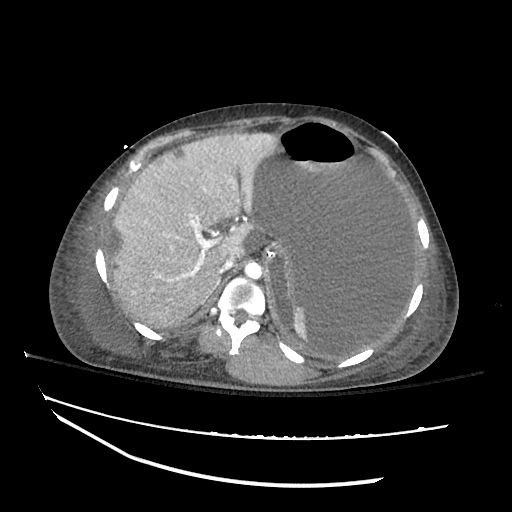
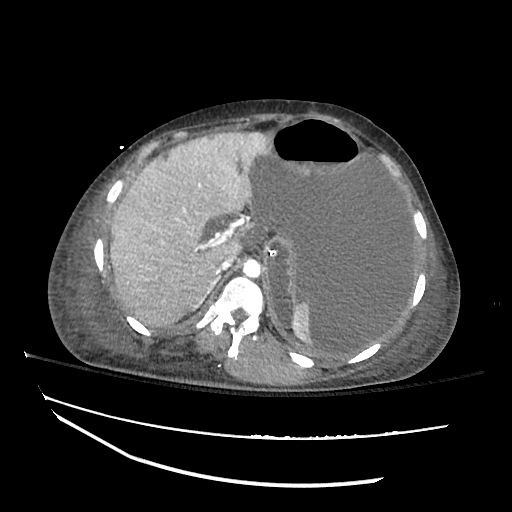
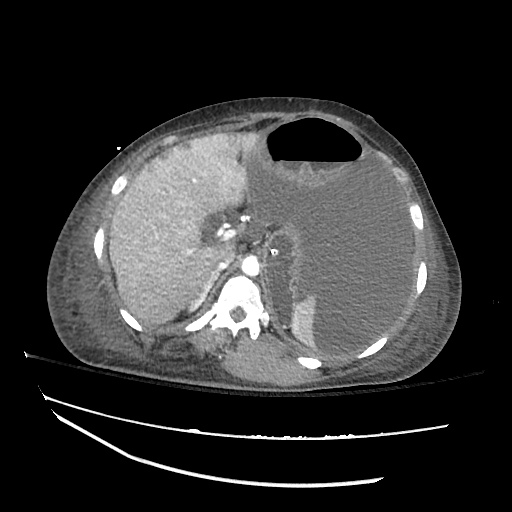
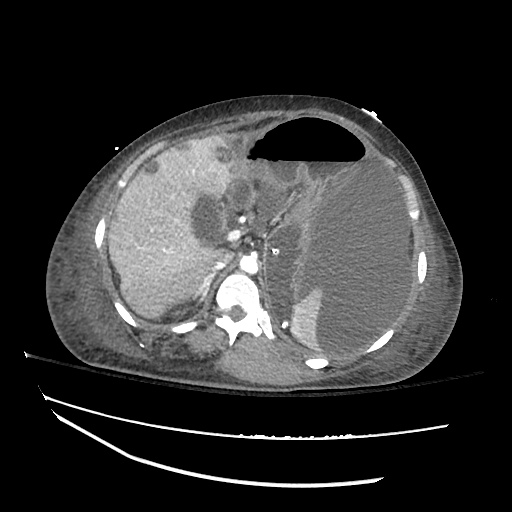
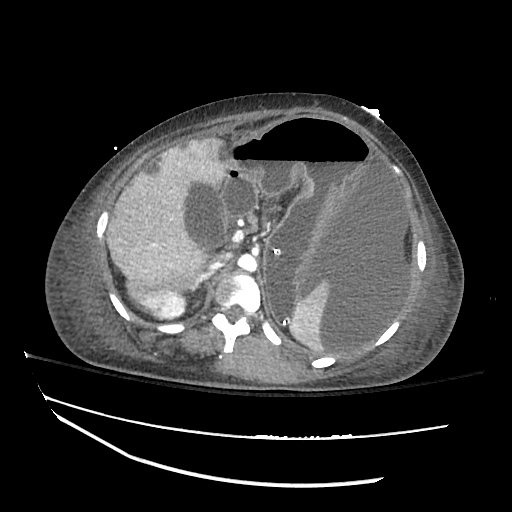
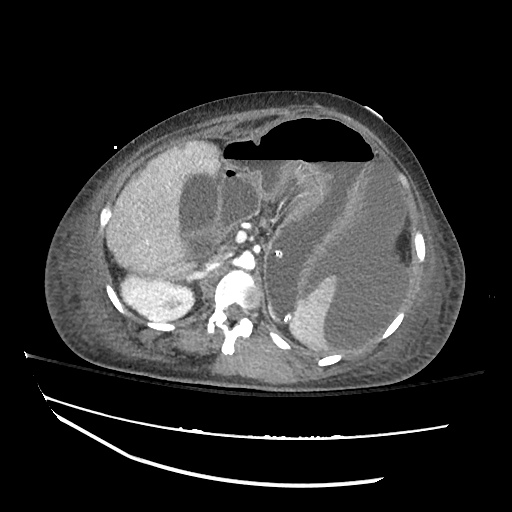
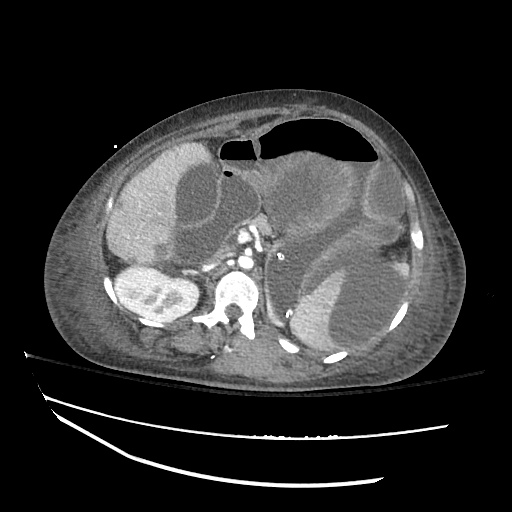
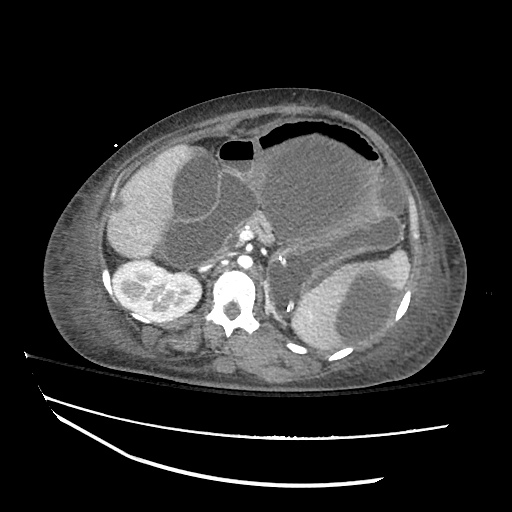
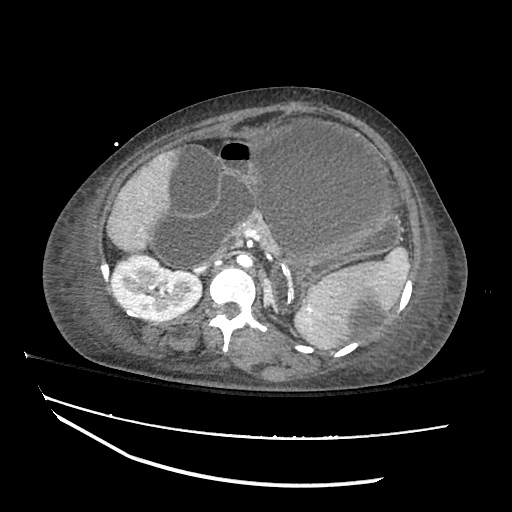
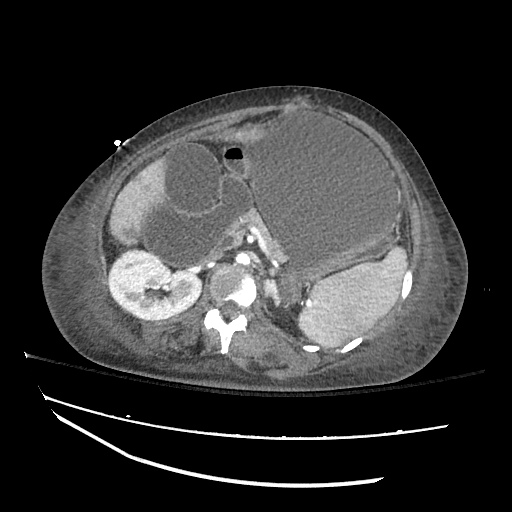
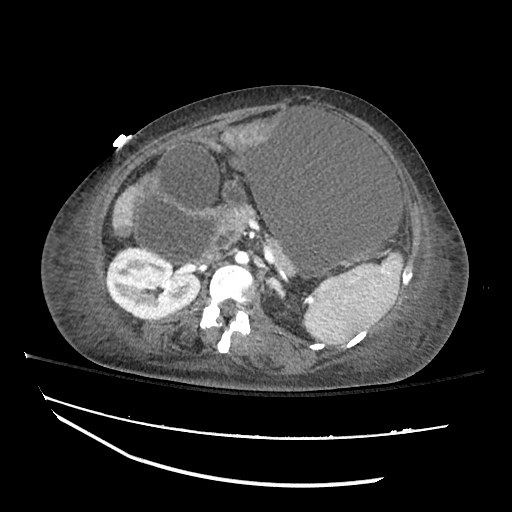
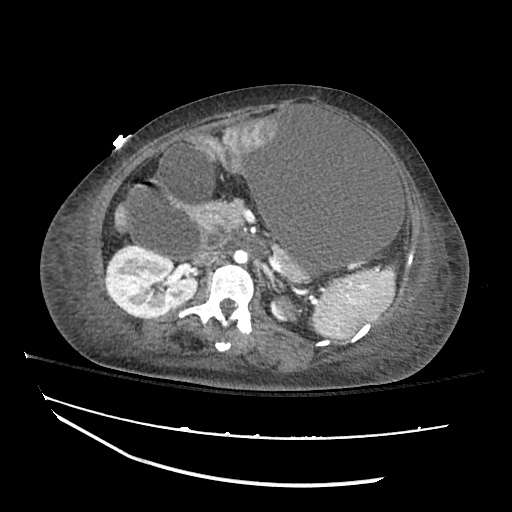
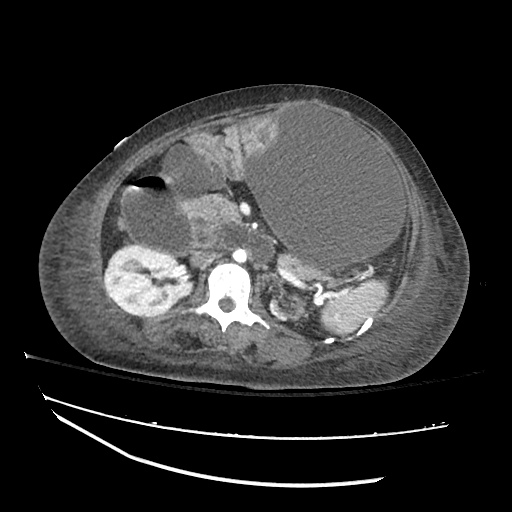
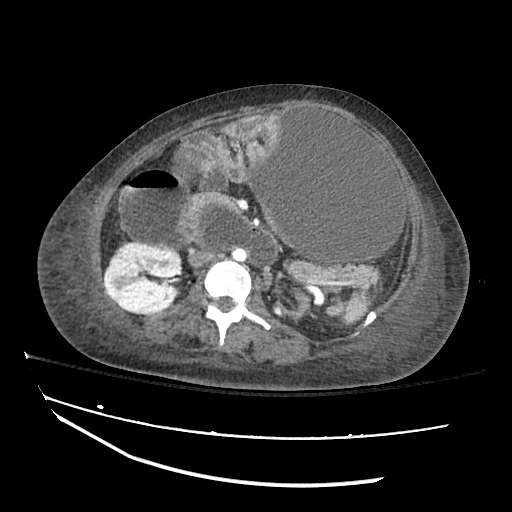
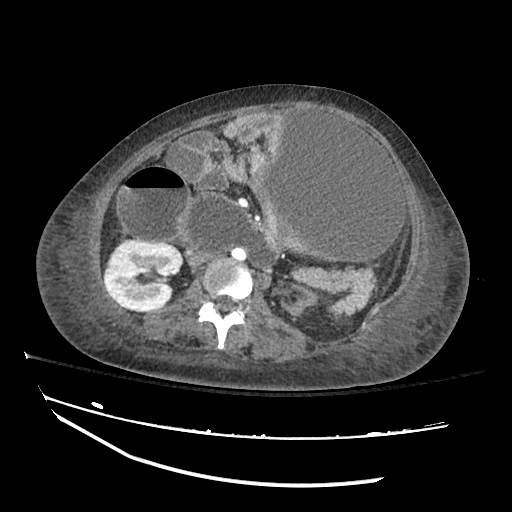
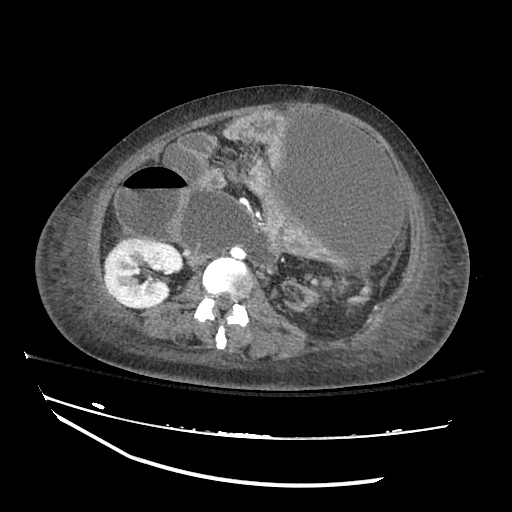
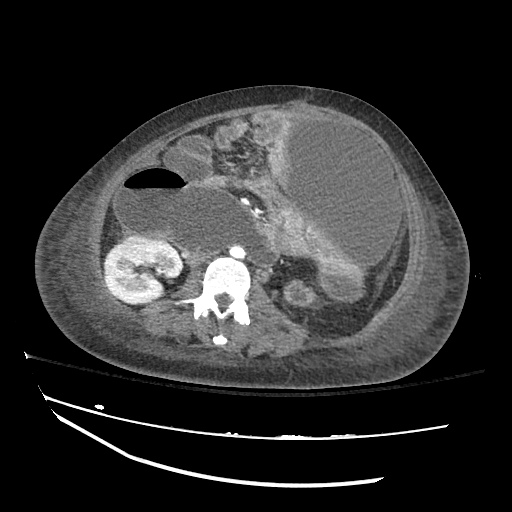
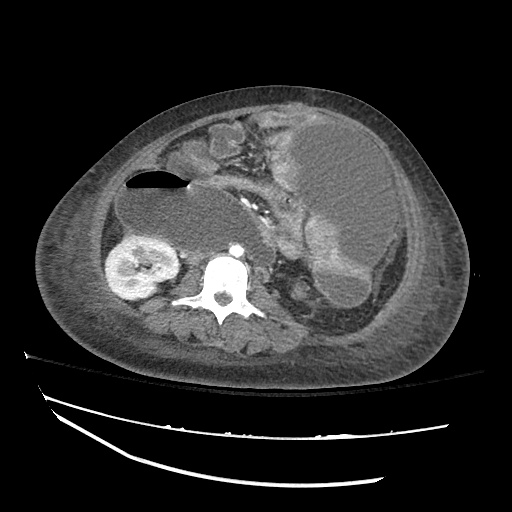
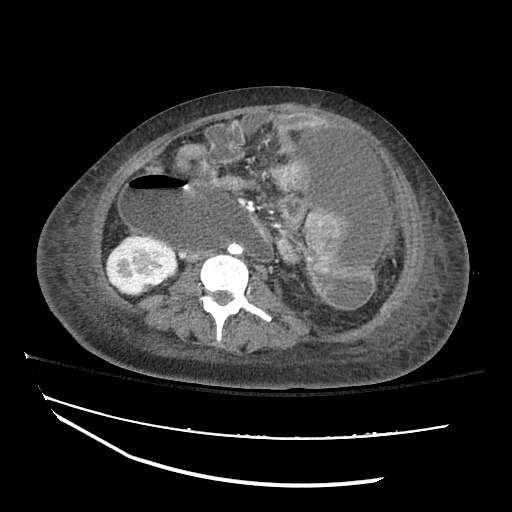
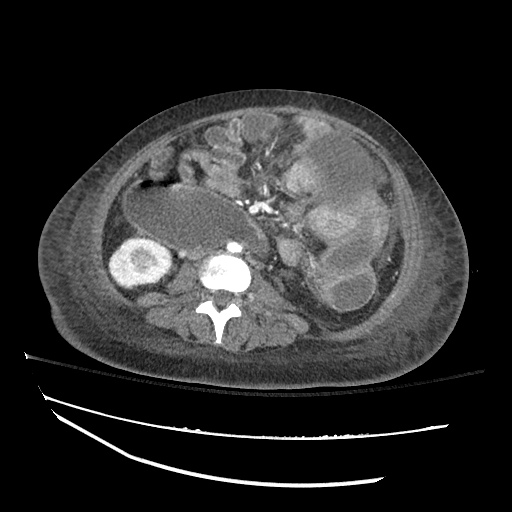
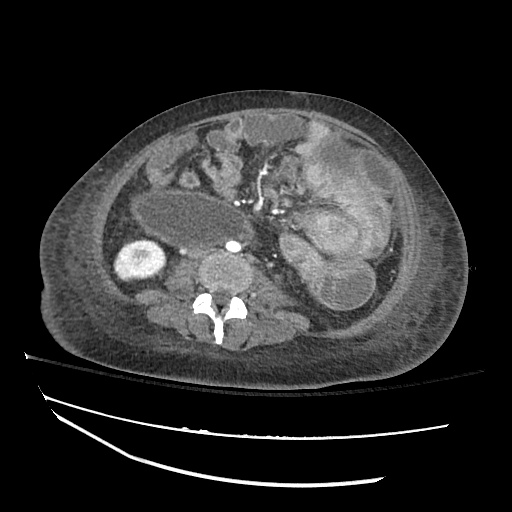
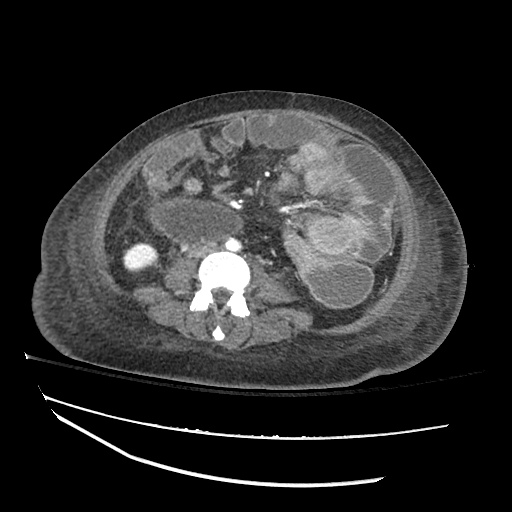
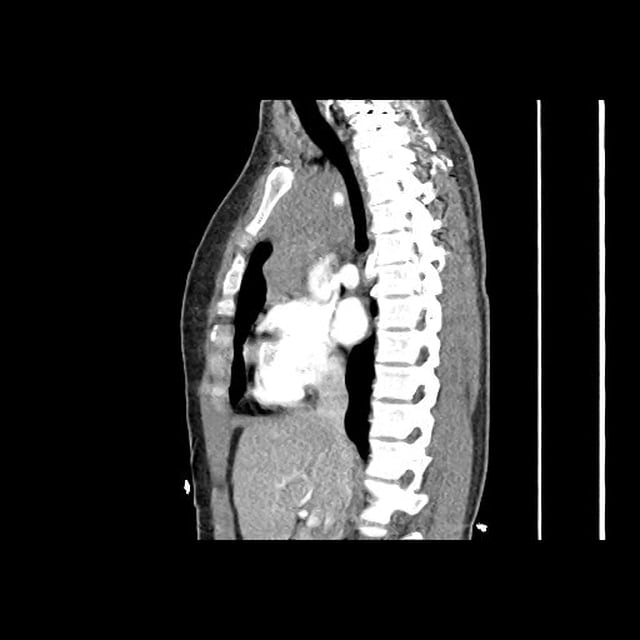
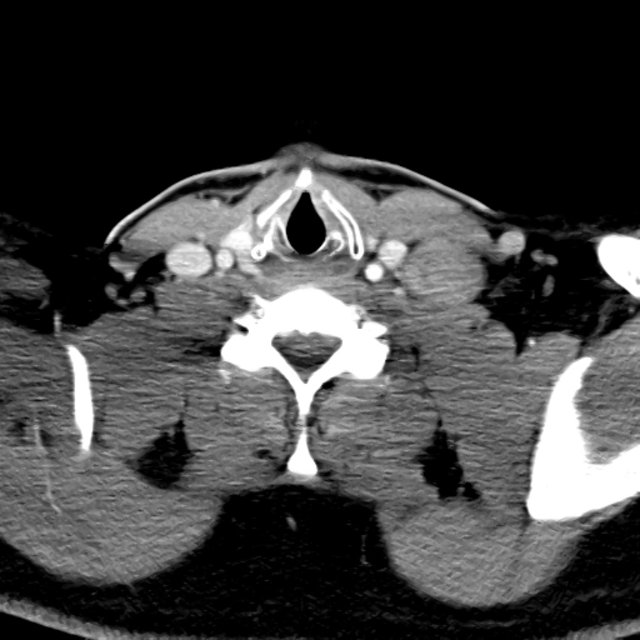
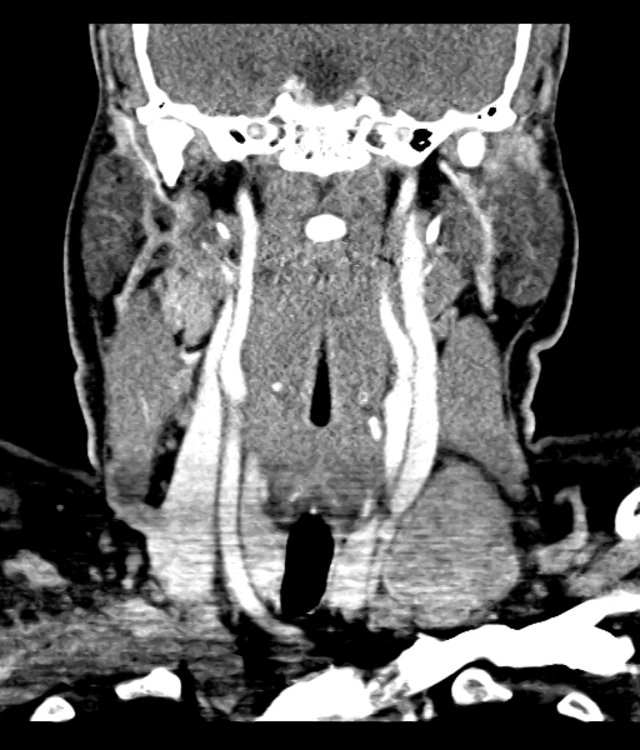
CT Abdomen/Pelvis
Assessment and Plan:
52yo G1P1 with likely primary ovarian adenocarcinoma with extensive peritoneal involvement, complicated by malignant ascites and pleural effusions with hypoxemia. The primary concern in this post-operative patient with a history of malignancy is venous thromboembolism, particularly pulmonary embolism. Aside from hypoxemia, this patient had no symptoms suggestive of pulmonary embolism (denied dyspnea, chest pain, cough, or lower extremity pain). Her examination had some signs infrequently associated with pulmonary embolism which were otherwise adequately explained by known bilateral pleural effusions (including decreased breath sounds, rales, and tachypnea), she was not tachycardic, had no evidence of jugular venous distension, nor a prominent P2. In addition, she was receiving appropriate VTE prophylaxis (both pharmacological and mechanical). The application of the Modified Wells clinical decision rule suggests low likelihood of pulmonary embolism (with 2.5 points assigned for recent surgery and history of malignancy). The patient had a recent CT chest without evidence of pulmonary embolism, and given that an elevated D-dimer was virtually assured which would have necessitated repeat imaging (and the associated risks of radiation exposure and contrast injury), the episode was ascribed to ventilation/perfusion mismatch secondary to large pleural effusions. Hypoxemia resolved after several minutes at rest and no further testing was performed.
Signs and Symptoms of Pulmonary Embolism1
| Signs | Symptoms |
|---|---|
| Tachypnea | Dyspnea (rest/exertion) |
| Tachycardia | Pleurtic chest pain |
| Rales | Cough |
| Decreased breath sounds | Orthopnea |
| Prominent P2 | Calf/thigh pain or swelling |
| JVD | Wheezing |
Modified Wells Criteria1,2
| Feature | Points |
|---|---|
| Clinical symptoms of DVT (leg swelling, tenderness to palpation) | 3.0 |
| Pulmonary embolism most likely diagnosis | 3.0 |
| Tachycardia (HR >100) | 1.5 |
| Immobilization >3d, surgery in prior 4 weeks | 1.5 |
| Prior DVT/PE | 1.5 |
| Hemoptysis | 1.0 |
| Malignancy | 1.0 |
>4.0 Likely
≤4.0 Unlikely
Algorithm for Evaluation of Suspected Pulmonary Embolism2
- Utility of D-Dimer:2
- Of limited utility in patients with high suspicion for pulmonary embolism
- Decreased specificity: malignancy, hospitalized patients, pregnancy, elderly
- Efficacy of DVT prophylaxis:3
- LMWH prevents approximately ½ of VTE events (including PE, symptomatic and asymptomatic DVT)
References:
- Kruip, M. J. H. A., Söhne, M., Nijkeuter, M., Kwakkel-Van Erp, H. M., Tick, L. W., Halkes, S. J. M., Prins, M. H., et al. (2006). A simple diagnostic strategy in hospitalized patients with clinically suspected pulmonary embolism. Journal of internal medicine, 260(5), 459–466. doi:10.1111/j.1365-2796.2006.01709.x
- Tapson, V. F. (2008). Acute pulmonary embolism. The New England journal of medicine, 358(10), 1037–1052. doi:10.1056/NEJMra072753
- Själander, A., Jansson, J.-H., Bergqvist, D., Eriksson, H., Carlberg, B., & Svensson, P. (2008). Efficacy and safety of anticoagulant prophylaxis to prevent venous thromboembolism in acutely ill medical inpatients: a meta-analysis. Journal of internal medicine, 263(1), 52–60. doi:10.1111/j.1365-2796.2007.01878.x